VIDEO
+3
Loading...
Cat. #BK036

Kit contents (20-80 assays)
Equipment & materials required
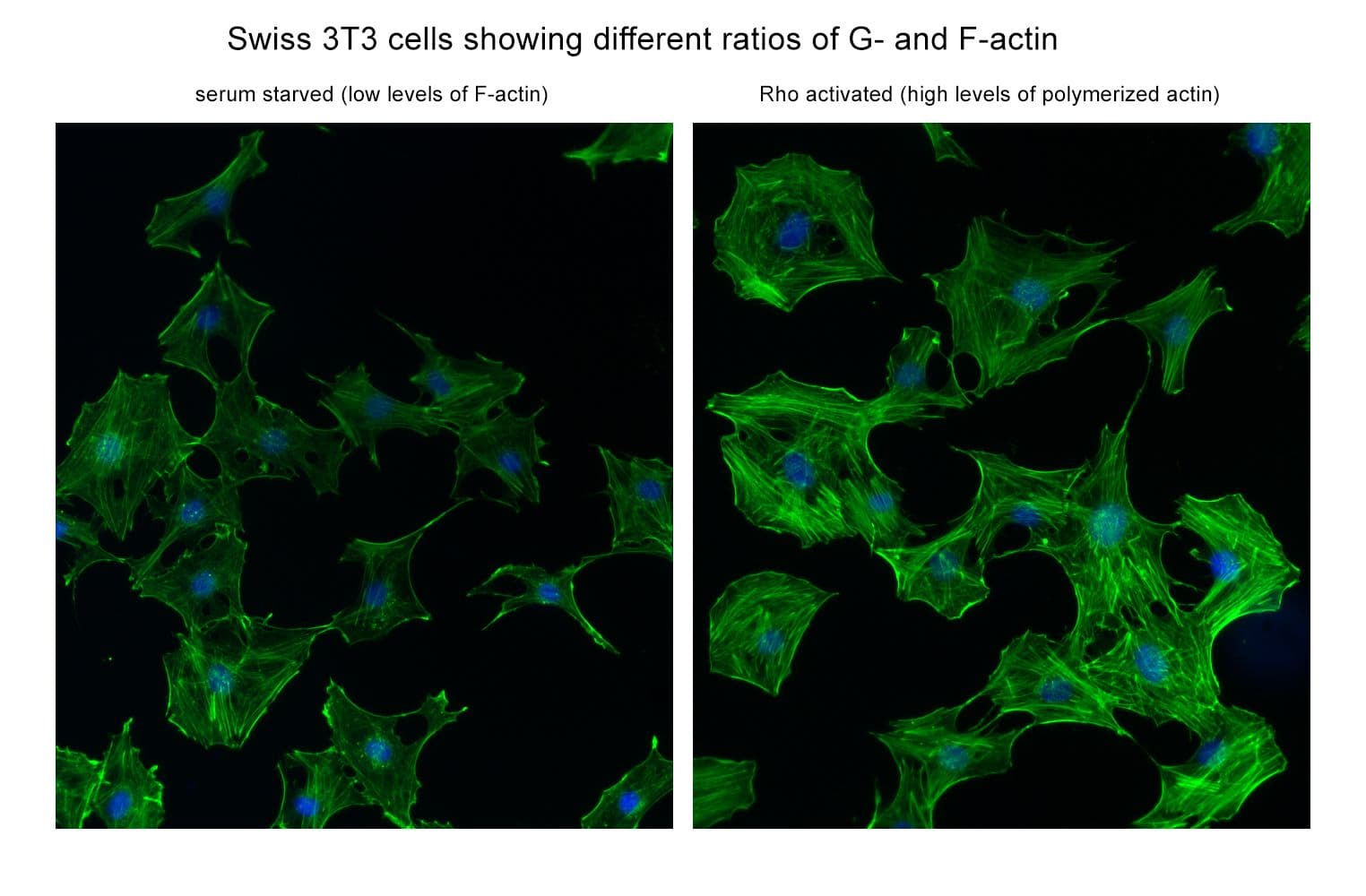
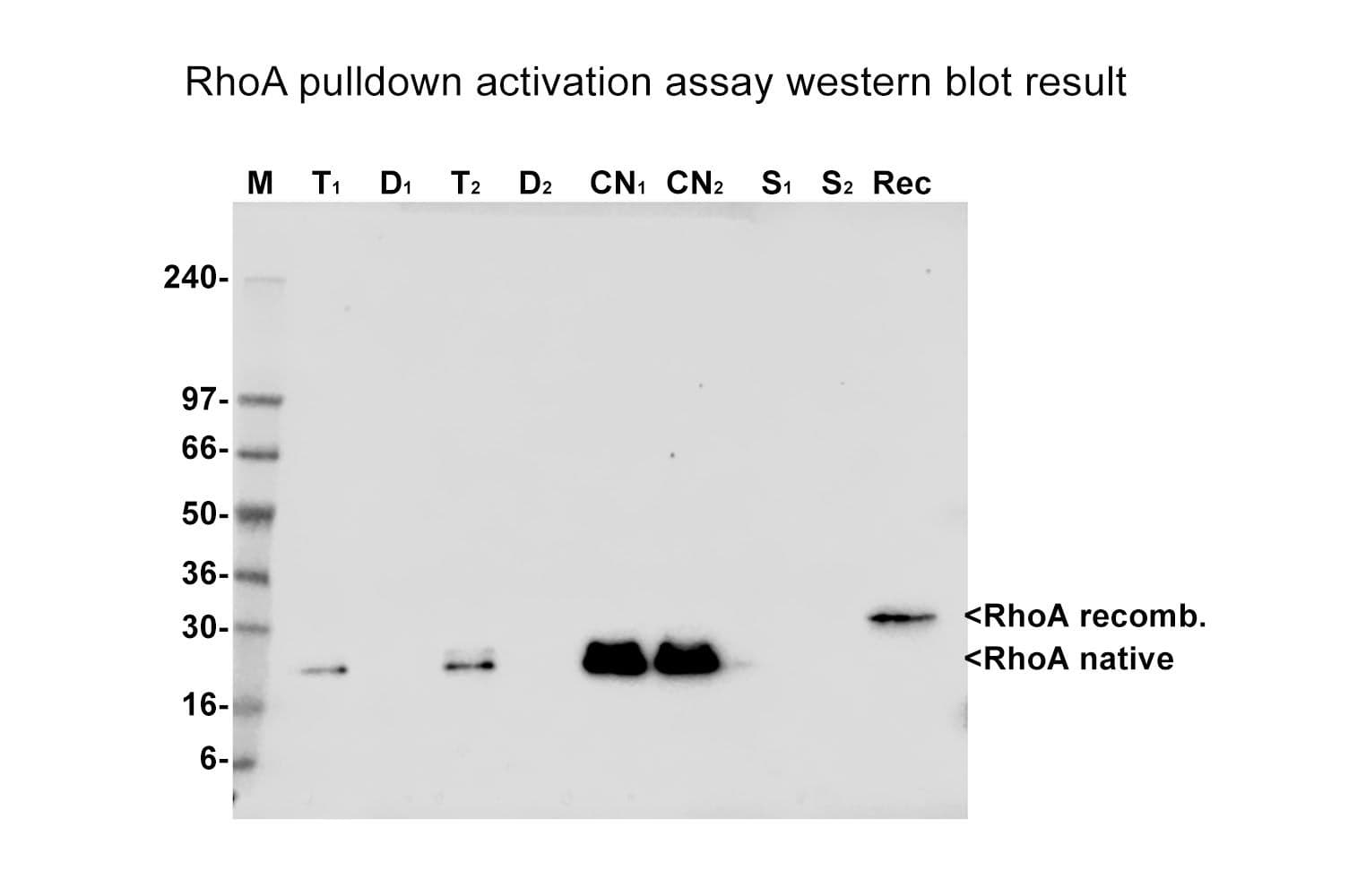
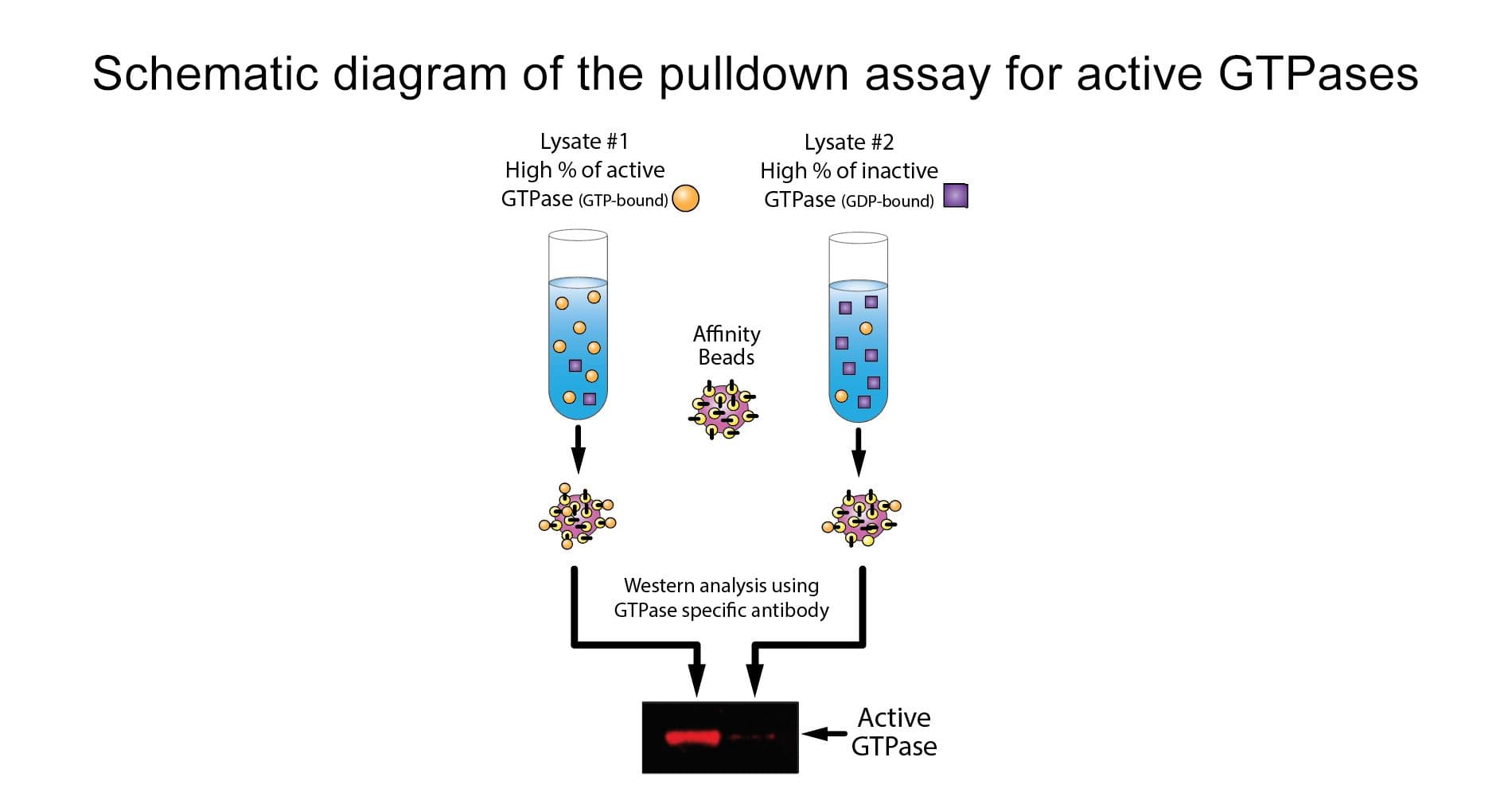
The RhoA pull-down assay isolates active RhoA (GTP-bound form) using the RhoA/B/C-binding domain (RBD) of an effector protein immobilized on agarose beads. The bound RhoA is then detected and quantified by Western blotting using a RhoA-specific antibody.
Key characteristics