Ras-related C3 botulinum toxin substrate 2 (Rac2) is a 21 kDa GTPase encoded by 192 amino acids that acts as a molecular switch in a variety of cellular events that include cell motility, development, signaling, cytoskeletal organization, release of inflammatory modulators, and innate immunity. Along with the Rho isoforms and Cdc42, it is a member of the Rho sub-family of Ras super-family GTPases and is one of 3 Rac isoforms (Rac1 and Rac3 being the others). Rac2 has approximately 88% sequence identity with Rac1 and Rac3. The three proteins diverge primarily in the last 15 residues of the C-termini. Biochemically, Rac2 has slower nucleotide association and is more efficiently activated by the Rac GEF Tiam1. Rac2’s functionality relies upon C-terminal lipid modifications, specifically modification by the C20 GG isoprenoid which mediates Rac2’s subcellular localization and association with membranes. Upon activation, Rac2 is mainly localized to endo-membranes.
Like all Ras super-family GTPases, Rac2 is bound to GTP in its active form, whereas it is inactive in its GDP-bound form. Cycling between an active and inactive state is governed by guanidine activating proteins (GEFs) that exchange bound GDP for GTP and GTPase-activating proteins (GAPs) that promote GTP hydrolysis. Several GEFs have been shown to activate Rac2 selectively: Vav1, Tiam1, P-Rex1, Swap70, and Dock2. Two GAPs that target Rac2 are Abr and Bcr. In its GTP-bound, active state, Rac2 interacts with downstream effector proteins such as p67phox and cytochrome b-558, PLCbeta2, nitric oxide synthase 2 (NOS2), and Pak1.
Expression is restricted to hematopoietic cells with the highest expression in myeloid cells. During the differentiation of hematopoietic and myeloid cells, Rac2 expression is regulated. As might be expected, Rac2 has a specialized role in many hematopoietic and immunological processes. Rac2-deficient mice show defects in stem cells and mast cells, as well as B- and T-cells. Indeed, Rac2 is essential for integrin-mediated stem cell adhesion, as well as growth and migration. As expected, loss of Rac2 results in impairment in several actin-based functions and structures. There is consensus that Rac2 is the predominant Rac GTPase functioning in neutrophils as deletion of Rac2 (Rac2 -/- mice) results in decreased neutrophil motility and adhesion, major defects in cortical F-actin assembly, and accordingly, chemotaxis and reduced phagocytosis and superoxide production by NAPH oxidase. B-cell development is also impaired in Rac2 -/- mice. For T-cell differentiation, Rac2 activity is required for interferon-gamma production both in vitro and in vivo during normal T-cell activation, and in Th1-cell differentiation, through simultaneous activation of both the NFkappaB and p38 pathways. In mast cells, loss of Rac2 results in defects in growth, survival, chemotaxis, and adhesion, as well as degranulation. Rac2 is critical in regulating the growth factor-induced survival through activation of Akt and a change in expression levels of the Bcl-2 family members BAD and Bcl-XL.
Rac2 is implicated in a variety of diseases, including primary immunodeficiency syndrome (not surprising given the necessity of Rac2 for neutrophil function) and multiple cancers (e.g., brain cancer, head and neck squamous cell cancer, acute myeloid leukemia [AML], and chronic myelogenous leukemia [CML]).
Images of Rac2
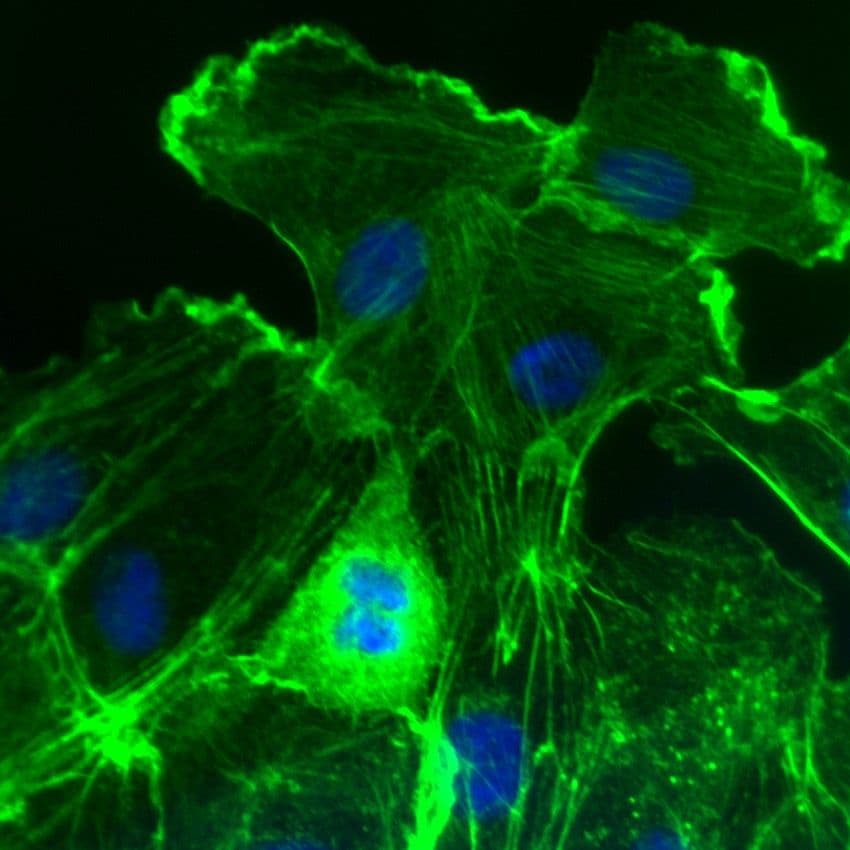
For more information about Rac2, please see here:
Fritz G. and Henninger C. 2015. Rho GTPases: Novel players in the regulation of the DNA damage response? Biomolecules. 5, 2417-2434.
Duquette P.M. and Lamarche-Vane N. 2014. Rho GTPases in embryonic development. Small GTPases. 5, 8.
Lin Y. and Zheng Y. 2015. Approaches of targeting Rho GTPases in cancer drug discovery. Expert Opin. Drug Discov. 10, 991-1010.
Carstanjen D. et al. 2005. Rac2 regulates neutrophil chemotaxis, superoxide production, and myeloid colony formation through multiple distinct effector pathways. J. Immunol. 174, 4613-4620.
Gomez del Pulgar T. et al. 2005. Rho GTPase expression in tumourigenesis: evidence for a significant link. Bioessays. 27, 602-613.
Haeusler L.C. et al. 2003. Comparative functional analysis of the Rac GTPases. FEBS Lett. 555, 556-560.
Hwang S.L. et al. 2005. Rac2 expression and mutation in human brain tumors. Acta Neurochir (Wien). 147, 551-554.
Gomez del Pulgar T. and Lacal J.C. 2009. RAC2 (ras-related C3 botulinum toxin substrate 2 (rho family, small GTP binding protein Rac2)). Atlas Genet. Cytogenet. Oncol. Haematol. 13, 493-497.
Rac2 Related Products
Rac1 protein: His tagged: human wild type (Cat. # RC01)
Rac2 protein: His tagged: human wild type (Cat. # RC02-A)
Rac1 G-LISA Activation Assay (Luminescence format) - 96 assays (Cat. # BK126)
Rac1 G-LISA Activation Assay (Luminescence format) - 96 assays (Cat. # BK126)
Rac1 G-LISA Activation Assay Kit (Colorimetric Based) 96 assays (Cat. # BK128)
Anti-Rac1: mouse Mab (Cat. # ARC03)
SiR-Actin Kit 50 nmol SiR-Actin and 1 umol verapamil (Cat. # CY-SC001)
Tiam1 GEF Protein (DHPH Exchange Domain, aa1040-1406, MBP tag) (Cat. # CS-GE04)
Acti-stain 488 phalloidin (Cat. # PHDG1-A)
Acti-stain 555 phalloidin (Cat. # PHDH1-A)
Acti-stain 670 phalloidin (Cat. # PHDN1-A)
Rhodamine Phalloidin (Cat. # PHDR1)