Rac1 protein: GST tagged: human dominant negative
Product Uses Include
- Rac1 GEF binding studies
- Inhibition of Rac GEFs in vitro
- Inhibition of Rac proteins in vivo by microinjection
Material
The dominant negative form of the Rac1 protein contains a threonine to asparagine substitution at residue 17. The common name for this mutant is Rac1(T17N) (or N17Rac1). The asparagine substitution abolishes the protein's affinity for GTP and reduces its affinity for GDP. Hence, the Rac1(T17N) is always in either a nucletiode free state or in its inactive, GDP-bound, state. Because of this, it binds strongly to Rac GEFs and it blocks wild type Rac from being activated by these GEFs.
The dominant negative form of human Rac1 protein has been expressed in a bacterial system, and is available as a GST-tagged fusion protein. The recombinant protein is 50 kDa consisting of the Rac1 protein (22 kDa) and a 28 kDa GST tag. The tag is at the amino terminus of the protein. The protein is supplied as a lyophilized powder. When it is reconstituted in distilled water to 1 mg/ml, the protein is in the following buffer: 2 mM Tris pH 7.6, 0.5 mM MgCl2, 0.5% sucrose, 0.1% dextran. Protein concentration is determined by the Precision Red Advanced Protein Assay Reagent (Cat # ADV02).
For other forms of Rac1 as well as many other purified small G-proteins, see our main small G-protein product page.
Purity
Purity is determined by scanning densitometry of proteins on SDS-PAGE gels. Samples are >90% purae, the major contaminant running at 28 kDa is GST protein.
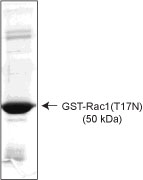
| Figure 1: GST-Rac1(T17N) purity determination. 10 µg of C17G01 was run on a SDS-PAGE gel and stained with Coomassie blue |
Biological Activity
Activity of Rac1(T17N) was verified by the ability of the protein to inhibit GEF activation of wild type Rac1 in an in vitro GEF assay (Cat # BK100).
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com

