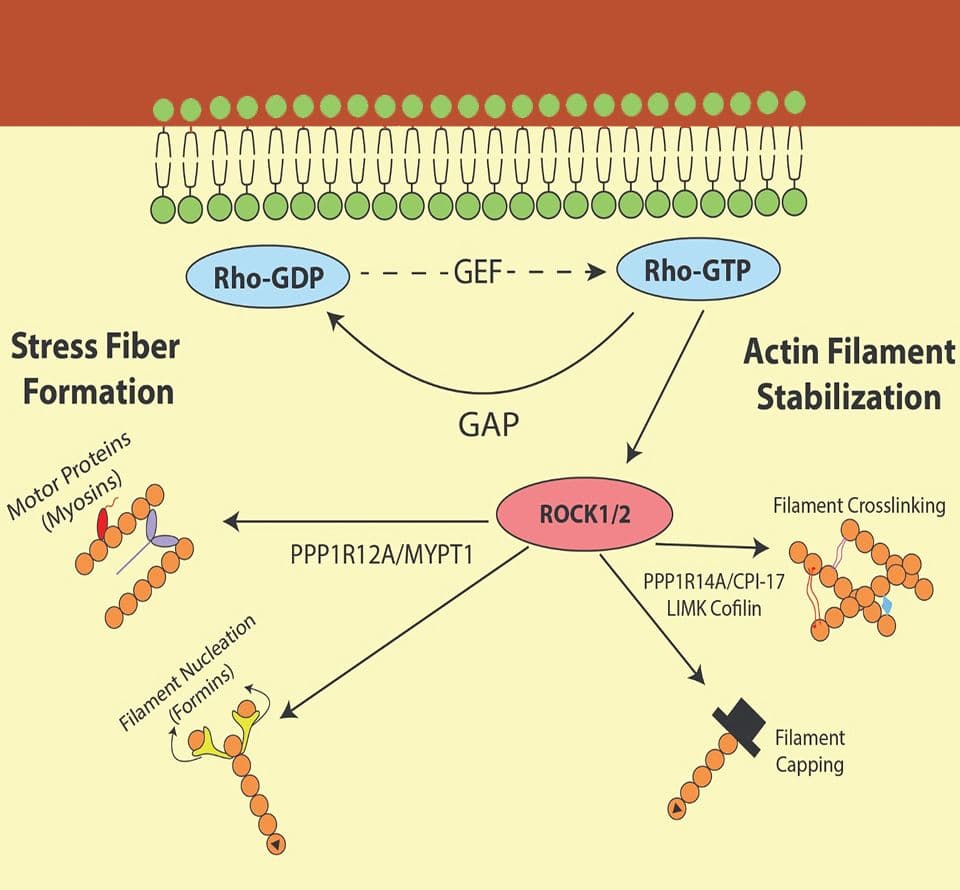
Small G-proteins
Cyto is the leading supplier of high-purity, biologically active Ras superfamily small GTPases.
Our extensive portfolio includes proteins in wild-type, constitutively active, and dominant negative forms, designed to support diverse research needs in signal transduction and cell biology.
Each GTPase product undergoes a rigorous four-point QC validation process, ensuring the highest quality standards:
- Accurate quantity per tube
- ≥90% purity
- Validated biological activity assay
- Western blot using the specific GTPase antibody
Trust Cyto for reliable, reproducible results in your GTPase-based research.
Cyto is the leading supplier of high-purity, biologically active Ras superfamily small GTPases.
Our extensive portfolio includes proteins in wild-type, constitutively active, and dominant negative forms, designed to support diverse research needs in signal transduction and cell biology.
Each GTPase product undergoes a rigorous four-point QC validation process, ensuring the highest quality standards:
- Accurate quantity per tube
- ≥90% purity
- Validated biological activity assay
- Western blot using the specific GTPase antibody
Trust Cyto for reliable, reproducible results in your GTPase-based research.