+3
Loading...
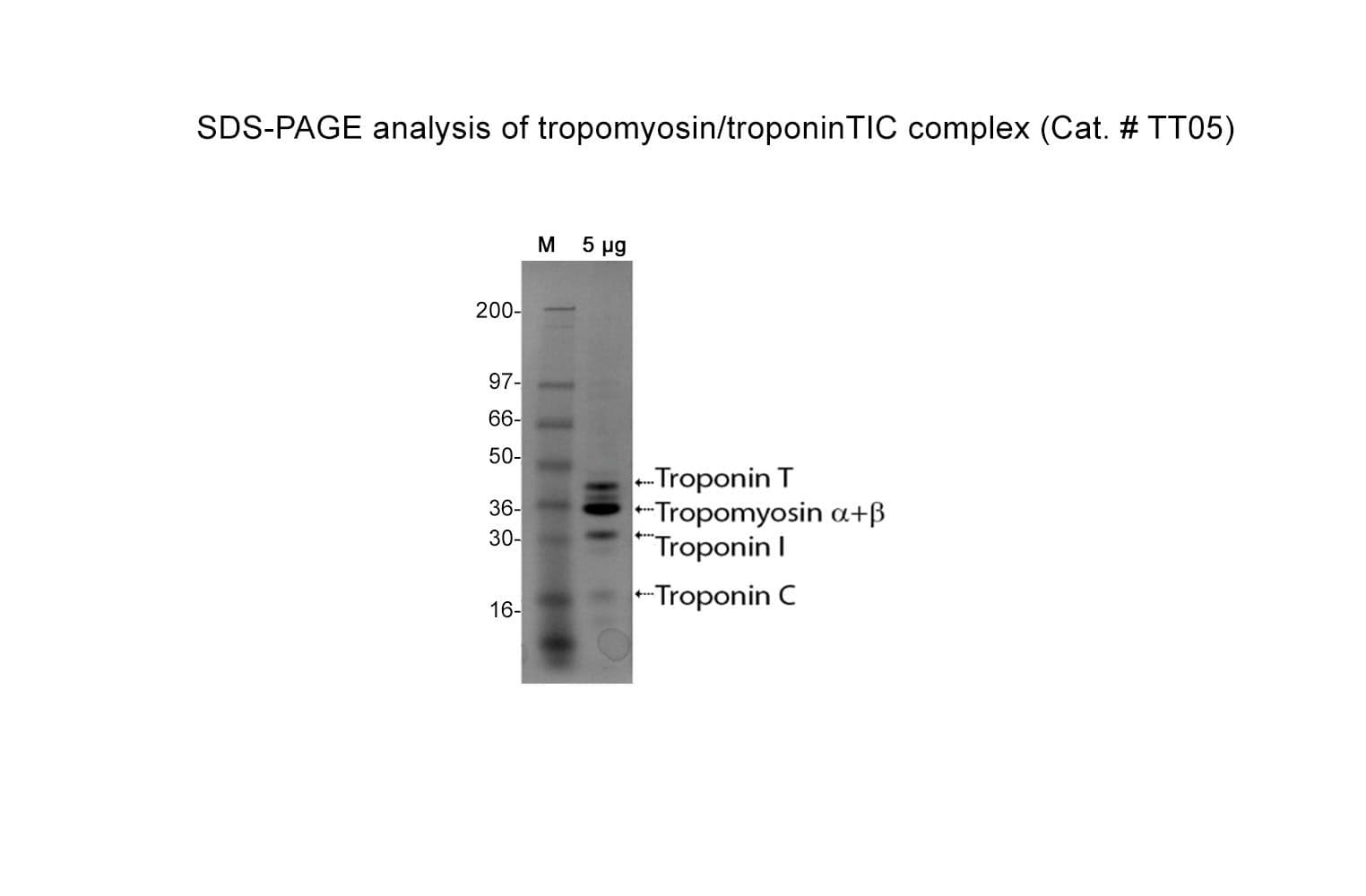
The Tropomyosin / Troponin protein (TT) complex has been purified from bovine cardiac muscle. The TT complex is composed of five proteins Tropomyosin α, Tropomyosin β, Troponin C, Troponin I, and Troponin T, in a stoichiometric ratio of 1:1:1:1:1. The complex is supplied as a white lyophilized powder.
Protein purity is determined by scanning densitometry of Coomassie Blue-stained protein on a 4-20% gradient polyacrylamide gel. The purity of the TT complex is >85%.
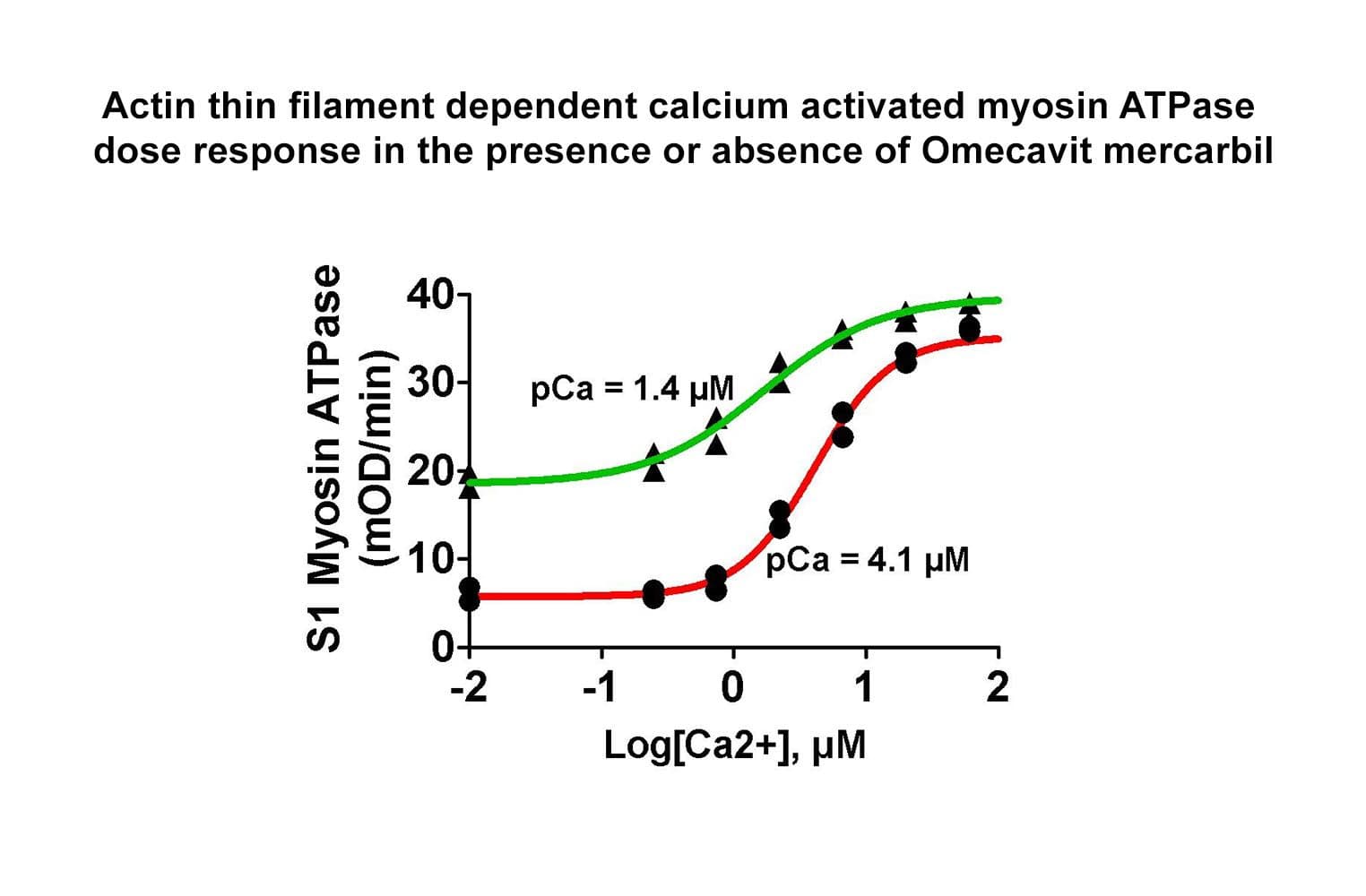
The biological activity of the TT complex is determined by measuring its regulation of F-actin–activated myosin ATPase.
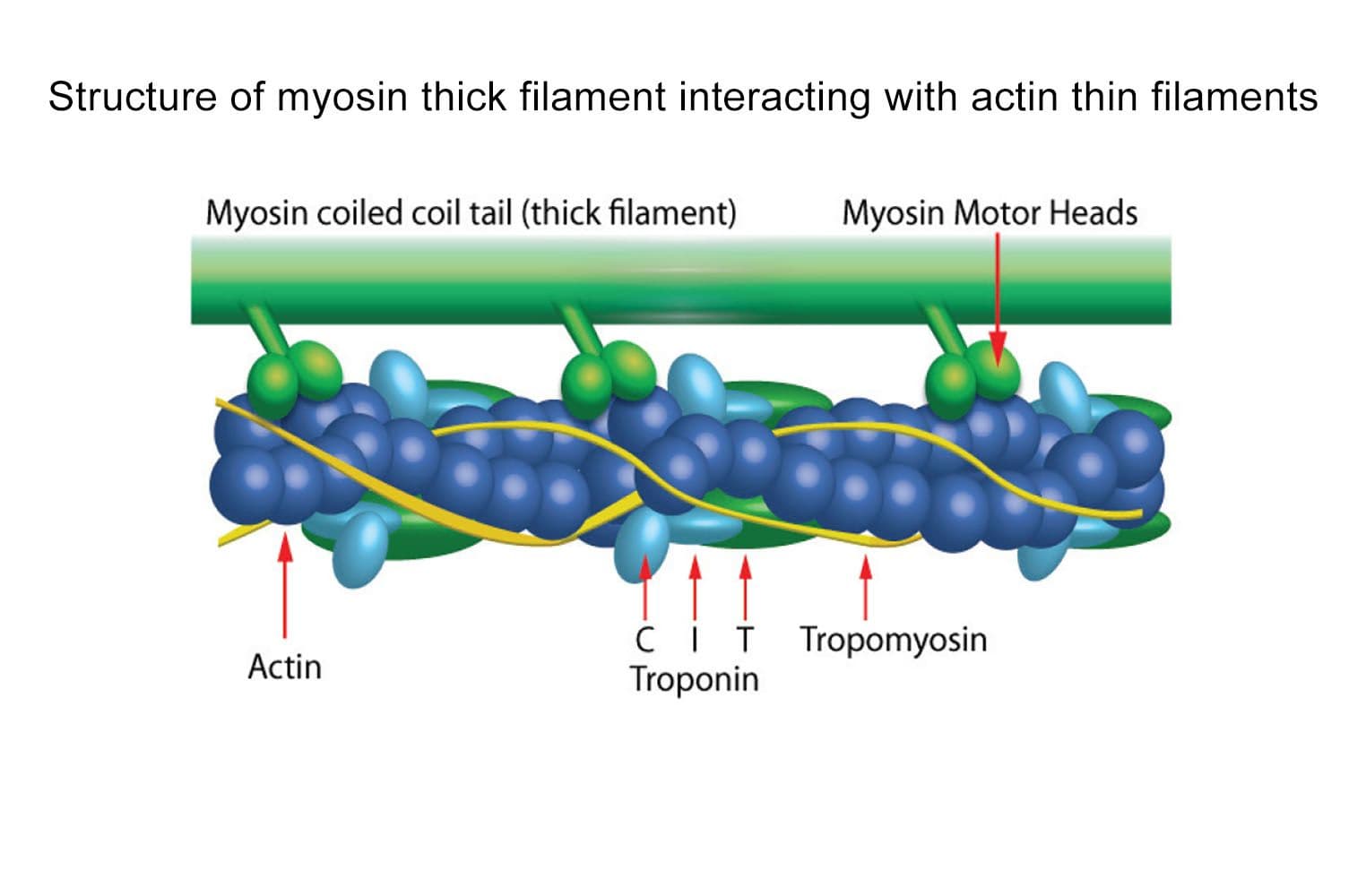
Mechanism:
Cat. #CS-TT05