BimC kinesin motor domain protein (KIF11): GST tagged: A. nidulans
Product uses
- Measurement of microtubule-activated ATPase activity
- Identification/characterization of proteins or small molecules that affect motor ATPase activity
- Identification/characterization of proteins or small molecules that affect kinesin motility
- Identification/characterization of proteins or small molecules that affect motor/microtubule interactions
Material
The conserved motor domain of Aspergillus nidulins BimC (KIF11) was expressed in a prokaryotic system. The recombinant protein contains a GST-Tag at the amino terminal end and has a combined molecular weight of 72 kD. The protein has been determined to be biologically active in a microtubule-activated ATPase activity test (see below). The protein is supplied as a lyophilized powder.
Purity
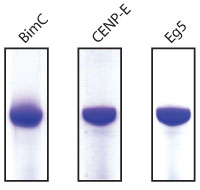
Protein purity is determined by scanning densitometry of Coomassie Blue stained protein on a 12% polyacrylamide gel. All kinesin motor domains are >85% pure. See figure 1 for example purities.
Biological Activity - Microtubule Activated ATPase Assay
BimC ATPase activity was measured by monitoring real time free phosphate generation using the Kinesin ELIPA Assay Kit (cat.# BK060). The assay is based upon an absorbance shift (330 nm - 360 nm) that occurs when 2-amino-6-mercapto-7-methylpurine ribonucleoside (MESG) is catalytically converted to 2-amino-6mercapto-7-methylpurine in the presence of inorganic phosphate (Pi). One molecule of Pi will yield one molecule of 2-amino-6mercapto-7-methylpurine in an essentially irreversible reaction. Hence, the absorbance at 360 nm is directly proportional to the amount of Pi generated in the kinesin ATPase reaction. Under the conditions outlined below, the Vmax for BimC microtubuleactivated ATPase activity for this Lot was 600 nmoles ATP generated per minute per mg of BM01 (Figure 2). The ATPase rate for this Lot using a 10 minute endpoint assay (Kinesin ATPase End Point Assay Kit, cat.# BK053) was 340 nmoles ATP per minute per mg of BM01. Both of these values are above the guaranteed minimum.
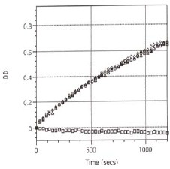
Figure 2. BM01 microtubule-activated ATPase activity using the Kinesin ELIPA Assay Kit (cat.# BK060).
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: What is the minimum concentration this kinesin motor protein can be stored at?
Answer 1: For storage at -70°C, the protein concentration should not be less than 5 mg/ml. Kinesin diluted below 5 mg/ml should not be re-frozen as it will lose activity.
Question 2: What is the most common use of this motor domain protein?
Answer 2: The BimC motor domain protein is most commonly used in conjunction with microtubules to measure microtubule-stimulated BimC ATPase activity. The ATPase activity can be measured with an endpoint or kinetic assay. Other uses of this motor protein include identification/characterization of proteins or small molecules that affect motor ATPase activity and identification/characterization of proteins or small molecules that affect motor/microtubule interactions.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com