BLOG
+3
Loading...
Cat. #CS-UPP01
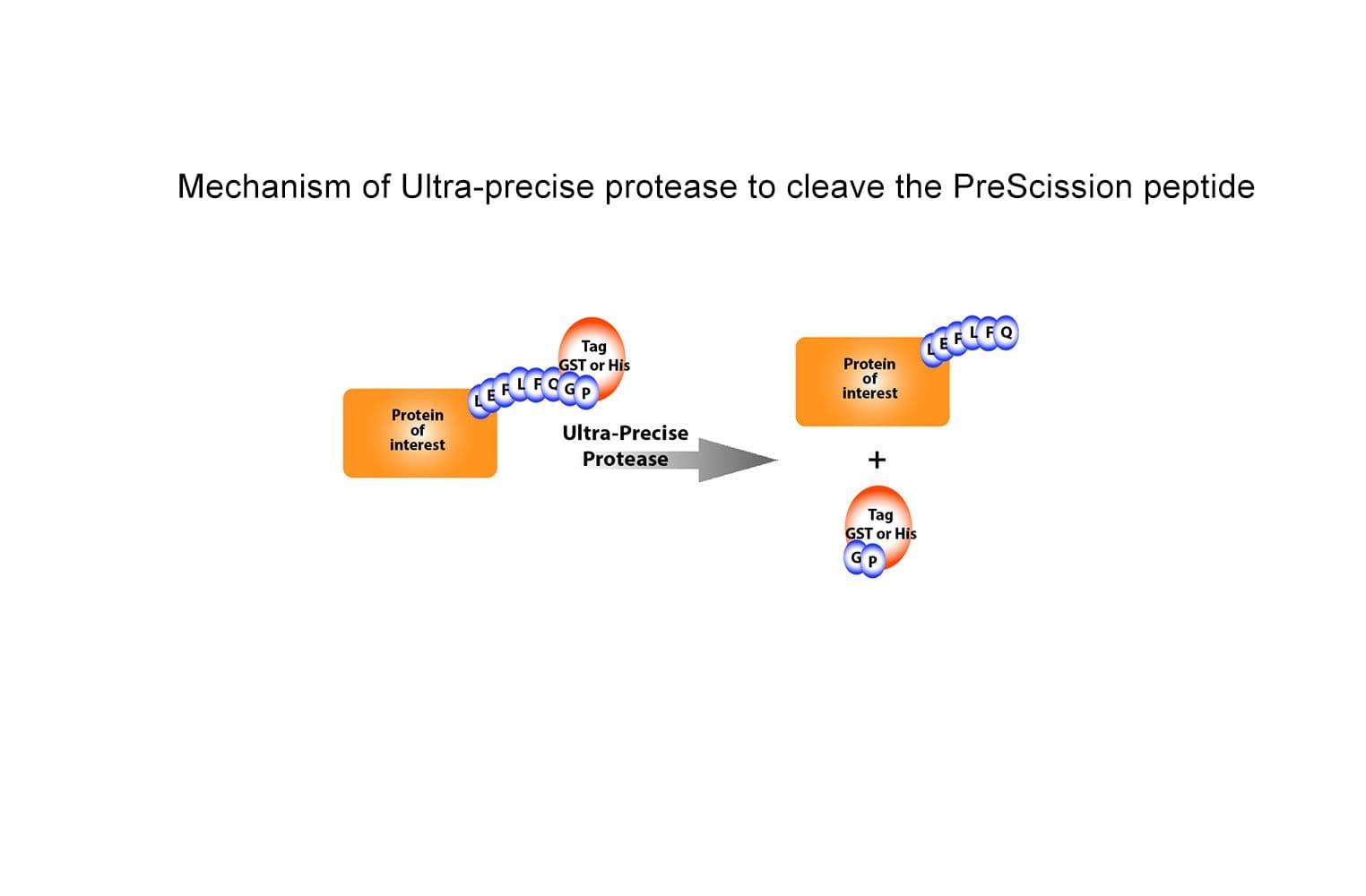

The Ultra-Precise HRV 3C Protease (UP protease) is a fusion protein with a glutathione S-transferase affinity tag (GST) and human rhinovirus (HRV) type 14 3C protease commonly used in molecular biology and protein purification techniques. It is identical in sequence and activity to the popular PreScission protease.
The recombinant UP protease protein was produced in a bacterial expression system.
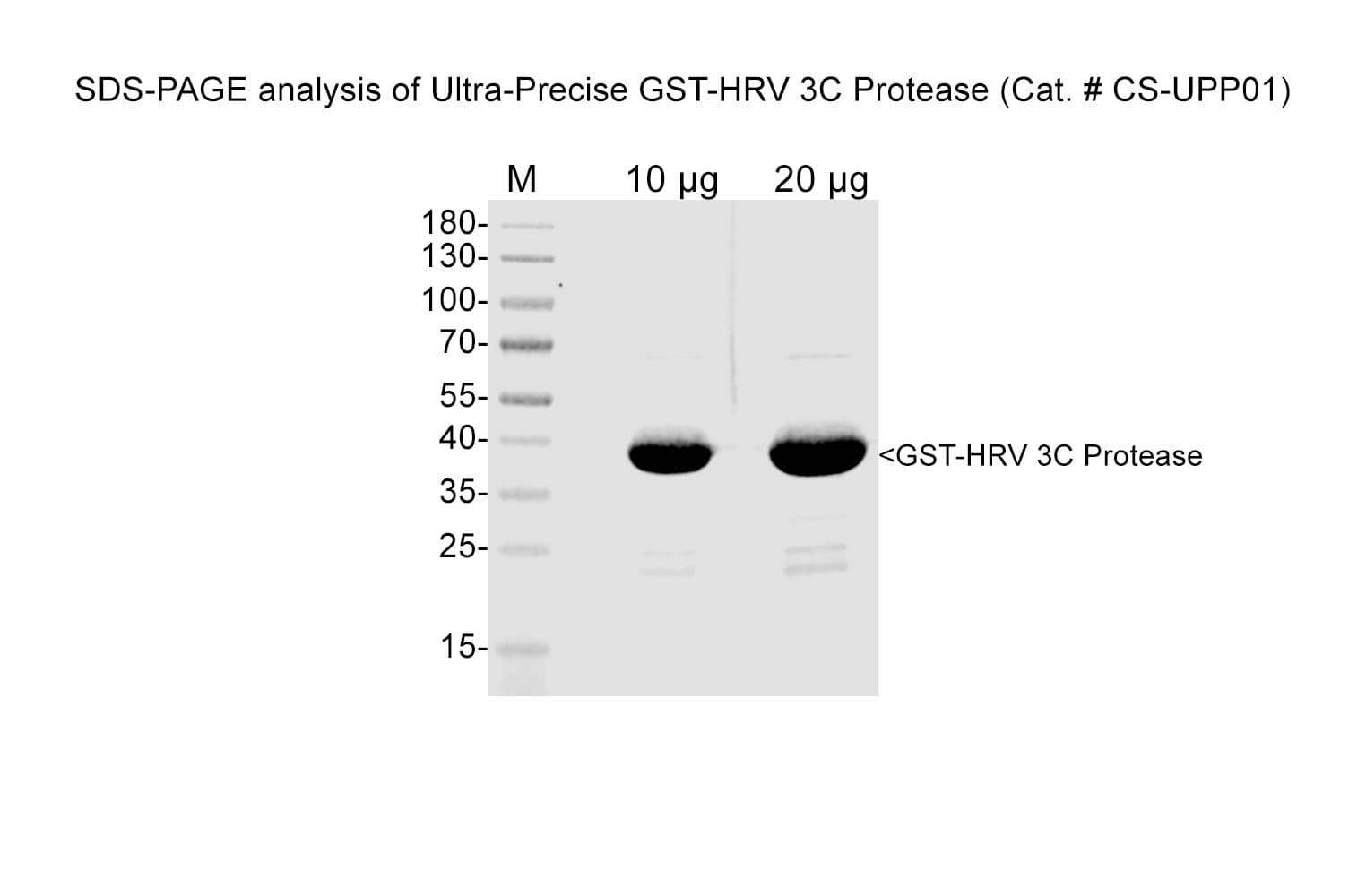
Protein purity is assessed by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity was determined to be ≥90% pure.
One unit is the amount of enzyme capable of cleaving >90% of 100 g of fusion protein in 16 hours at 4°C. Protease specific activity is 800-1100 units/mg.