WASP protein VCA domain: GST tagged: human
Product Uses Include
- Stimulating Arp2/3 complex activity
- Isolating Arp2/3 complex from cell or tissue extract
Material
The VCA (Verprolin, cofilin, acidic) domain of human WASP protein has been expressed in a bacterial expression system as a GST-tagged fusion protein. The protein has been purified by glutathione affinity chromatography and is supplied as a lyophilized powder. When reconstituted with distilled water, the complex is in the following buffer: 20 mM Tris pH 7.5, 25 mM KCl, 1 mM MgCl2, 0.5 mM EDTA, 0.2% dextran, 2% sucrose. The molecular weight of the GST-VCA domain protein is approximately 43 kDa.
Purity
Purity is determined by scanning densitometry of proteins on SDS-PAGE gels. Samples are >95% pure (Figure 1).
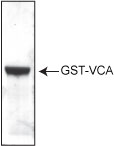
Figure 1: Purity determination of GST-WASP VCA domain. 20 µg of VCG03 was run on an SDS-PAGE gel and visualized by coomassie blue staining. Protein quantitation was performed using the Precision Red Protein Assay Reagent (Cat.# ADV02).
Biological Activity
VCG03 was tested in an actin polymerization assay (Cat. # BK003). In conjunction with Arp2/3 (Cat. # RP01) it was shown to stimulate actin polymerization 20-fold compared to the control without VCG03. This indicates VCA domain specificity for stimulating the actin nucleating activity of Arp2/3.
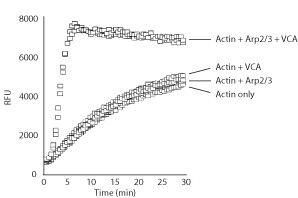
Figure 2: Actin polymerization stimulated by Arp2/3 complex and the VCA domain of WASP. Actin polymerization was measured using kit BK003. The addition of Arp2/3 complex or the VCA domain alone to actin has minimal effects on actin polymerization, while the combination of Arp2/3 and the VCA domain strongly stimulates the rate of actin polymerization.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: By itself, what is the effect of VCA-domain WASP protein on actin polymerization?
Answer 1: By itself, the VCA-domain WASP protein (Cat. # VCG03) partial inhibits actin polymerization. When combined with Arp2/3 (Cat. # RP01), actin polymerization is greatly enhanced. See the associated datasheets for details.
Question 2: What is the optimal concentration of Arp2/3 and VCA-domain WASP protein to modify actin polymerization?
Answer 2: We recommend that pyrene actin is present at a final concentration of 0.8 μM, Arp2/3 complex at 10 nM (Cat. # RP01) and the VCA domain of WASP at 400 nM (Cat. # VCG03). The critical factor here is the actin concentration which must be 0.80 +/-0.2 µM in the final reaction. This concentration is just below the critical concentration for assembly and hence will not polymerize on it’s own.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com






