TECH GUIDE
+3
Loading...
Cat. #BK011P


Kit contents (sufficient for 96 assays)
Equipment & materials required
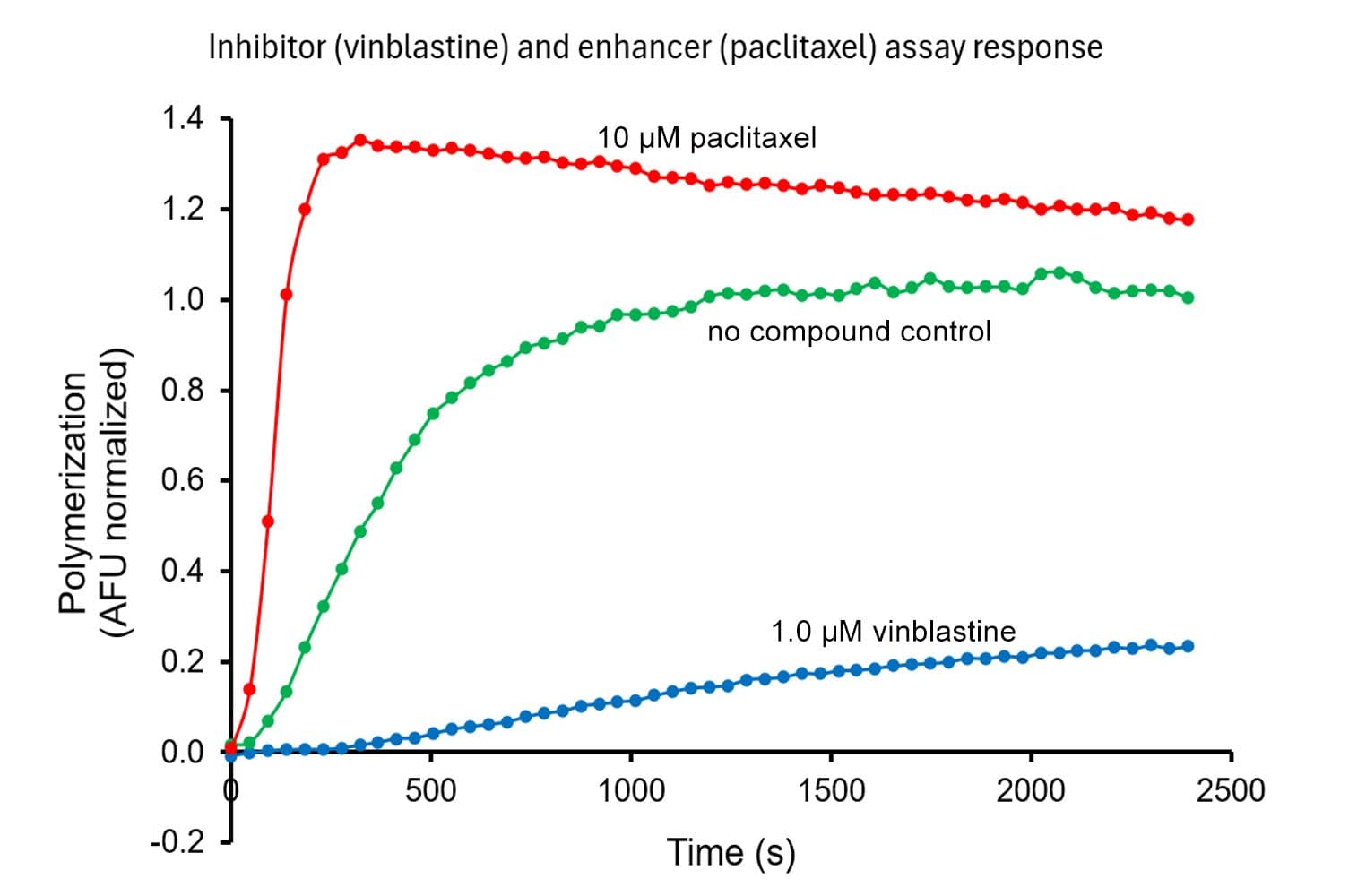
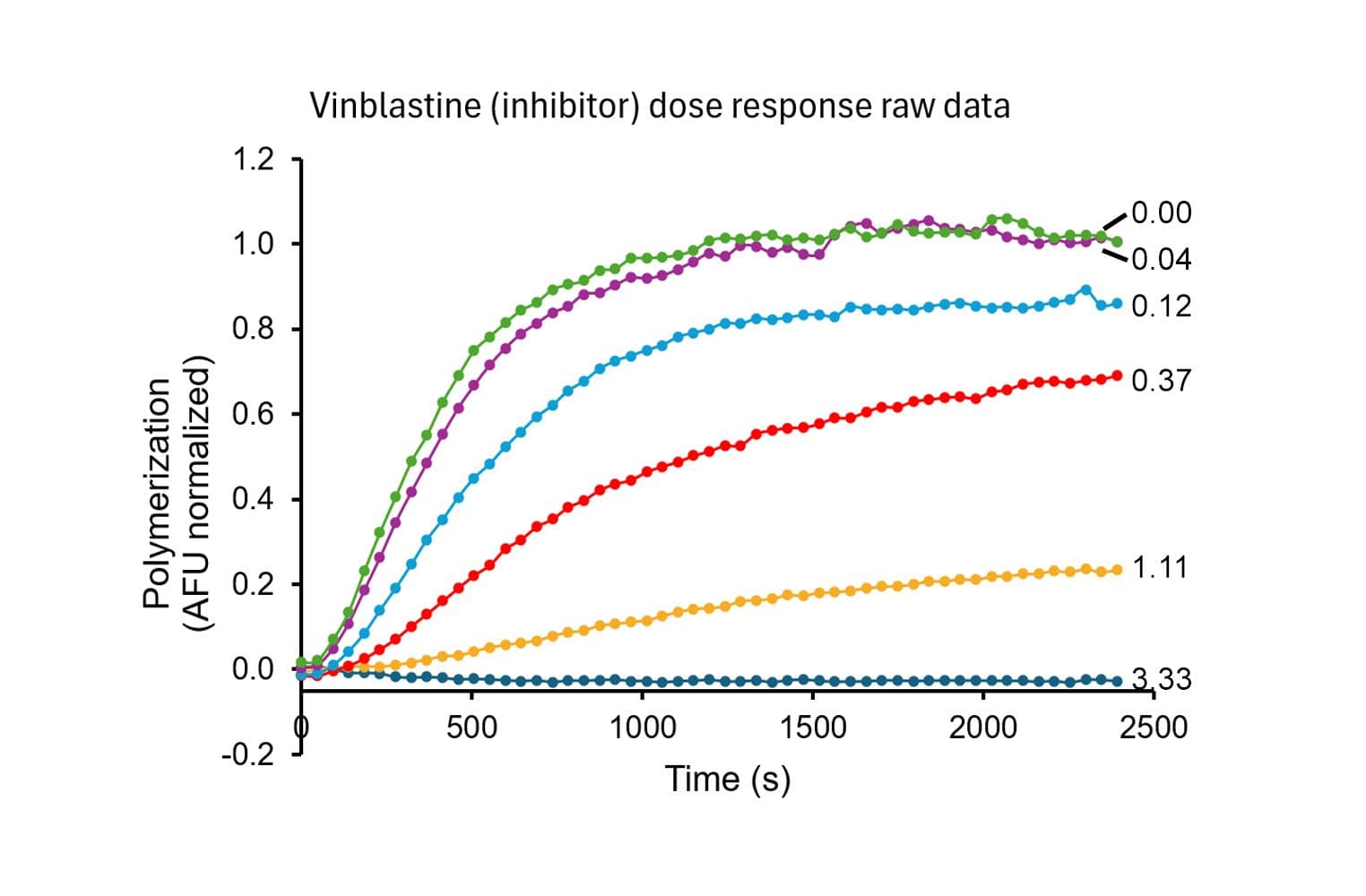
This assay measures tubulin polymerization dynamics using a fluorescent reporter that incorporates into microtubules as they form. It is designed to monitor the real-time kinetics of microtubule assembly and disassembly, which is critical for understanding cellular functions and screening pharmacological agents.
Key features