Plexin-B2 orchestrates collective stem cell dynamics via actomyosin contractility, cytoskeletal tension and adhesion
- By Cytoskeleton Inc. - Live Cell News
- Feb 17, 2022

Multicellular organization requires rapid changes in biomechanical dynamics and establishment of a collective actomyosin network in order to maintain cell morphology. How human embryonic stem cells (hESCs) orchestrate cytoskeletal tension across cells and organize adhesion complexes to maintain cytoarchitectural integrity is incompletely understood. Recent work by the Friedel lab identified a key role for Plexin-B2 in the control of actomyosin contractility and adhesion properties in hESCs. Plexin-B2 is an axon guidance receptor that has been shown to mediate cellular interactions in a variety of tissue contexts. The group utilized Plexin-B2 CRISPR KO hESCS (B2-KO) as well as lentiviral Plexin-B2 overexpressing hESCs [B2-OE] to show that dysregulation of Plexin-B2 severely alters hESC colony expansion and geometry. Utilizing RNA-Seq approaches they identified pathways related to muscle contraction, ECM organization, integrin signaling, and elastic fiber formation to be particularly augmented. A deeper investigation into the morphological changes revealed significant alterations in both F-actin networks and phosphorylated myosin light chain 2 localization. The dysfunction in actin networks were verified in live cells using SYP555-actin in combination with live-cell imaging, which highlighted a sluggish reorganization of F-actin in B2-OE cells, underscoring a temporal element at play. Atomic force microscopy studies performed by the group support the role of Plexin-B2 in controlling hESC colony formation through an actomyosin coordinated mechanism. Additionally, they determined that Plexin-B2 also affect cell-cell and cell-matrix adhesion properties. Utilizing multiple molecular and pharmacological approaches they identified downstream mediators such as RAP1/2 effectors, calcium activation, and the YAP mechanosensor as critical intermediary players by which Plexin-B2 controls cytoskeletal tension and adhesion in hESCs. Cytoskeleton’s live cell actin probe (Cat. # CY-SC202) and their MemGlow membrane probes were employed in this study to help reveal the cytoskeletal machinery utilized by Plexin-B2 to control morphogenesis.
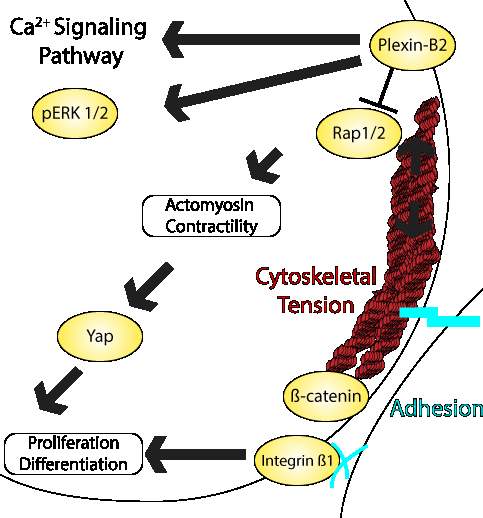
Above Schematic: Plexin-B2 signaling with mechanoregulation in stem cells
Link to Citation:
Related Products/Products Used
Spy-555 Actin (Cat. # CY-SC202)
MemGlow™ 488: Fluorogenic Membrane Probe (A Member Of The MEMBRIGHT™ Family) (Cat. # MG01)
