Product Uses Include
- Observation of fibronectin matrix assembly (1)
- Cell invasion assays
- Bioengineering; study of cell adhesion to polymers (2)
Material
Fibronectin is purified from bovine plasma. Protein purity is determined by scanning densitometry of coomassie blue stained protein on a 4-20% polyacrylamide gel. Biotinylated fibronectin is >80% pure (Figure 1).
The protein is modified to contain covalently linked biotins at random surface lysines. A long-chain activated ester of biotin [biotin-XX, succinimidyl ester] is used to label the protein. Labeling efficiency is determined by the ability to detect 10 ng biotinylated fibronectin using alkaline phosphatase conjugated streptavidin (Figure 2).
Fibronectin runs as individual subunits on SDS-PAGE with an apparent molecular weight of 230 kDa. FNR03 is supplied as a white lyophilized powder. Each vial contains 20 µg protein.
Purity
Purity is determined by scanning densitometry of proteins on SDS-PAGE gels. Samples are >80% pure.
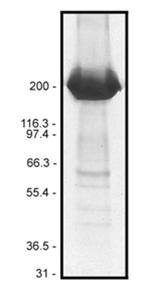
Figure 1: Fibronectin Purity Determination
Legend: 50 µg of biotinylated fibronectin was separated by electrophoresis in a 4-20% SDS-PAGE system. The protein was stained with coomassie blue. Protein quantitation was determined with the Precision Red™ Protein Assay Reagent (Cat. # ADV02). Mark12 molecular weight markers are from Invitrogen.

Figure 2: Detection of Biotinylated Fibronectin
Legend: Serial dilutions of biotinylated fibronectin were separated by electrophoresis on a 4-20% polyacrylamide gel, blotted to PVDF, probed with a 1:1000 dilution of streptavidin alkaline phosphatase (Sigma) and detected with 1-Step NBT/BCIP reagent™ (Pierce). Lane 1, 100 ng, Lane 2, 50 ng, Lane 3, 40 ng, Lane 4, 20 ng, and Lane 5, 10 ng of biotinylated fibronectin. Lane M, SeeBlue™ molecular weight markers (Invitrogen). Arrow indicates biotinylated fibronectin.
References
- Pankov R and Yamada KM. 2004. Non-radioactive quantification of fibronectin matrix assembly. Curr. Protocols in Cell Biol. S25, 10.13.1—10.13.9.
- Lehnert M, et al. 2011. Adsorption and conformation behavior of biotinylated fibronectin on streptavidin-modified TiO(X) surfaces studied by SPR and AFM. Langmuir, 27, 7743-7751
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: What is the size of the spacer between the fibronectin and the biotin label?
Answer 1: The biotin label has been modified to contain a 14 atom spacer between the biotin and the fibronectin to improve the accessibility of biotin to avidin/streptavidin.
Question 2: What is the labeling efficiency of the biotinylated fibronectin?
Answer 2: Labeling efficiency is determined by the ability to detect 10 ng of biotinylated fibronectin using alkaline phosphatase-labeled streptavidin.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com


