Tubulin protein plus glycerol (>99% pure) Source: Sheep Brain
* Limited stock available. If stock is not available, Cytoskeleton will produce a new batch upon request. Minimum order will apply. Inquire for more information.
Product Uses
- IC50 & EC50 determinations for anti-tubulin ligands.
- Characterization of tubulin binding proteins
Background Information
Tubulin is composed of a heterodimer of two closely related 55 kDa proteins called alpha and beta tubulin. The two proteins are encoded by separate genes, or small gene families, whose sequences are highly conserved throughout the eukaryotic kingdom.
Tubulin polymerizes to form structures called microtubules (MTs). When tubulin polymerizes it initially forms protofilaments, MTs consist of 13 protofilaments and are 25 nm in diameter. Each µm of MT length is composed of 1650 heterodimers (1). Microtubules are highly ordered structures that have an intrinsic polarity (see Figure 1).
Figure 1. Microtubule Schematic
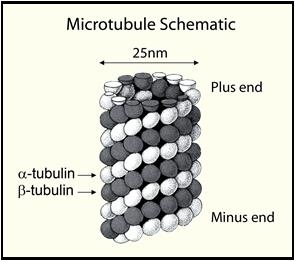
Tubulin can polymerize from both ends in vitro, however, the rate of polymerization is not equal. It has therefore become the convention to call the rapidly polymerizing end the plus -end and the slowly polymerizing end the minus-end. In vivo the plus-end of a microtubule is distal to the microtubule organizing center.
The intrinsic ability of pure tubulin to polymerize in vitro is very much a function of the experimental conditions. For example, one can manipulate the polymerization reaction to give microtubules of a particular mean length distribution or create conditions under which tubulin will not polymerize significantly until an enhancer component, such as a polymerization stimulating drug or protein, is added. The propensity of tubulin subunits to assemble into microtubules is dependent upon their affinity for microtubule ends (termed critical concentration [CC]). In order to achieve polymerization the CC needs to be less than the total tubulin concentration. At concentrations above the CC, tubulin will polymerize until the free subunit concentration is equal to the CC value. Because of this parameter, pure tubulin in General Tubulin Buffer (80 mM PIPES, pH 7.0, 2 mM MgCl2, 0.5 mM EGTA and 1 mM GTP) will not polymerize significantly at concentrations below 5 mg/ml. If, however, one adds a polymerization enhancer such as 5% glycerol, tubulin polymerization efficiency will approach 90% polymer mass at 37°C after 15-20 minutes. Tubulin polymerization is also a temperature sensitive event, optimal polymerization occurs at 37°C.
Materials
Tubulin protein has been purified from sheep brain by an adaptation of the method of Shelanski et al. (2). Further purification to >99% purity was achieved by cation exchange chromatography. Sheep brain tubulin is supplied in 0.10 ml aliquots at 12 mg/ml in 50 mM potassium MES pH 6.8, 0.25 mM MgCl2, 0.5 mM EGTA, 33% glycerol and 0.1 mM GTP. Tubulin consists of a heterodimer of one alpha and one beta isotype, each tubulin isotype is 55 kDa in size, SDS-PAGE analysis shows tubulin running as a 55 kDa species (see Figure 2). Typically, the molar equivalent of tubulin is defined as the heterodimer which has a molecular weight of 110 kDa.
Storage
It is recommended that T234S be stored at -70°C, where it is stable for 6 months. The protein should be rapidly thawed in a room temperature water bath, immediately transferred to ice and aliquoted into “experiment sized” amounts. Snap freeze aliquots in liquid nitrogen and store at -70°C. Aliquots of T234S MUST be snap frozen in liquid nitrogen prior to storage at -70°C, failure to do this results in significant loss of activity.
Purity
A 100 µg sample of T234S protein was separated by electrophoresis in a 4-20% SDS-PAGE system, and stained with Coomassie Blue. Purity of T234S was determined to be >99%. (Figure 2)
Figure 2. Purity Analysis of Tubulin Protein.
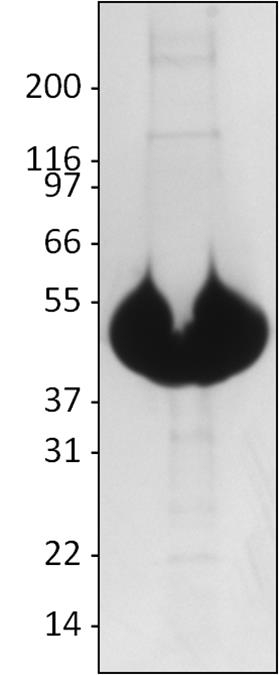
Legend: A 100 µg sample of T234S protein was separated by electrophoresis in a 4-20% SDS-PAGE system, and stained with Coomassie Blue. Protein quantitation was performed using the Precision Red Protein Assay Reagent (Cat. # ADV02). Molecular weight markers are from Invitrogen (Mark 12).
Note: Due to overloading of the gel, the tubulin band appears to run lower than the 55 kDa marker band.
Biological Activity Assay
The biological activity of T234S is assessed by a tubulin polymerization assay. The ability of tubulin to polymerize into microtubules can be followed by observing an increase in optical density of a tubulin solution at OD340nm (see Figure 3). Under the experimental conditions defined below a 6 mg/ml tubulin solution achieve an OD340nm absorbance reading between 1.14 - 1.56 per cm of light pathlength in 30 minutes at 37°C. The assay volume is 180 µl and assumes a spectrophotometer path-length of 0.8 cm, so the expected OD340nm is 0.91 to 1.25. NOTE: when using a microtiter plate compatible spectrophotometer the readings are taken from the top of the plate and therefore the volume of the reaction will directly influence the path-length. Cytoskeleton Inc. highly recommends the use of a 1/2 area well plate (Corning Cat. # 3696) for optimal polymerization signal in this assay.
Reagents
1. Tubulin protein (Cat. # T234S)
2. GTP stock (100 mM) (Cat. # BST06)
3. Custom 2x Tubulin Buffer; 80 mM PIPES pH 7.0, 8 mM MgCl2, 1 mM EGTA and 2 mM GTP
Equipment
1. Temperature regulated spectrophotometer set to 37°°C on kinetic mode at 340 nm.
2. Corning 96-well half area plates (Cat. # 3686) or other plate with low protein binding surface.
Method
1. Quickly thaw one tube of T234S in a room temperature waterbath and immediately transfer to ice.
2. Add 100 µl of Custom 2x Tubulin Buffer to the T234S in the microcentrifuge tube and mix well and keep on ice.
3. For a standard 96 well plate assay, transfer 180 ml of the Tubulin into a microtiter plate that has been pre-warmed to 37°°C. It is essential to use a 1/2 area 96-well plate for optimal signal generation in this assay. A minimum of 80 ul can be used for a 3 mm pathlength.
4. Measure tubulin polymerization by taking readings every 30 seconds at 340 nm and 37°C for 30 min to 1 h total.
5. Figure 2 shows the results of polymerizing T234S under the conditions described above.
Figure 3. Tubulin Polymerization Assay
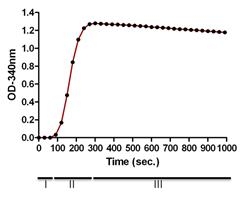
Legend: Polymerizations were carried out as indicated in the Method section. Polymerization was started by incubation at 37°C and followed by absorbance readings at 340 nm. Under these conditions polymerization reached a maximal OD340nm between 1.1-1.25 within 5 min, which is equivalent to >90% polymerization. The three phases of polymerization are shown, I (nucleation), II (growth), III (steady state).
For product Datasheets and MSDSs please click on the PDF links below.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com