Microtubule-binding proteins (MTBPs) regulate the dynamic behavior and stability of microtubules, essential for intracellular transport, cell shape, and division. By binding to tubulin or microtubule lattices, they control polymerization, depolymerization, and interactions with other cytoskeletal components.
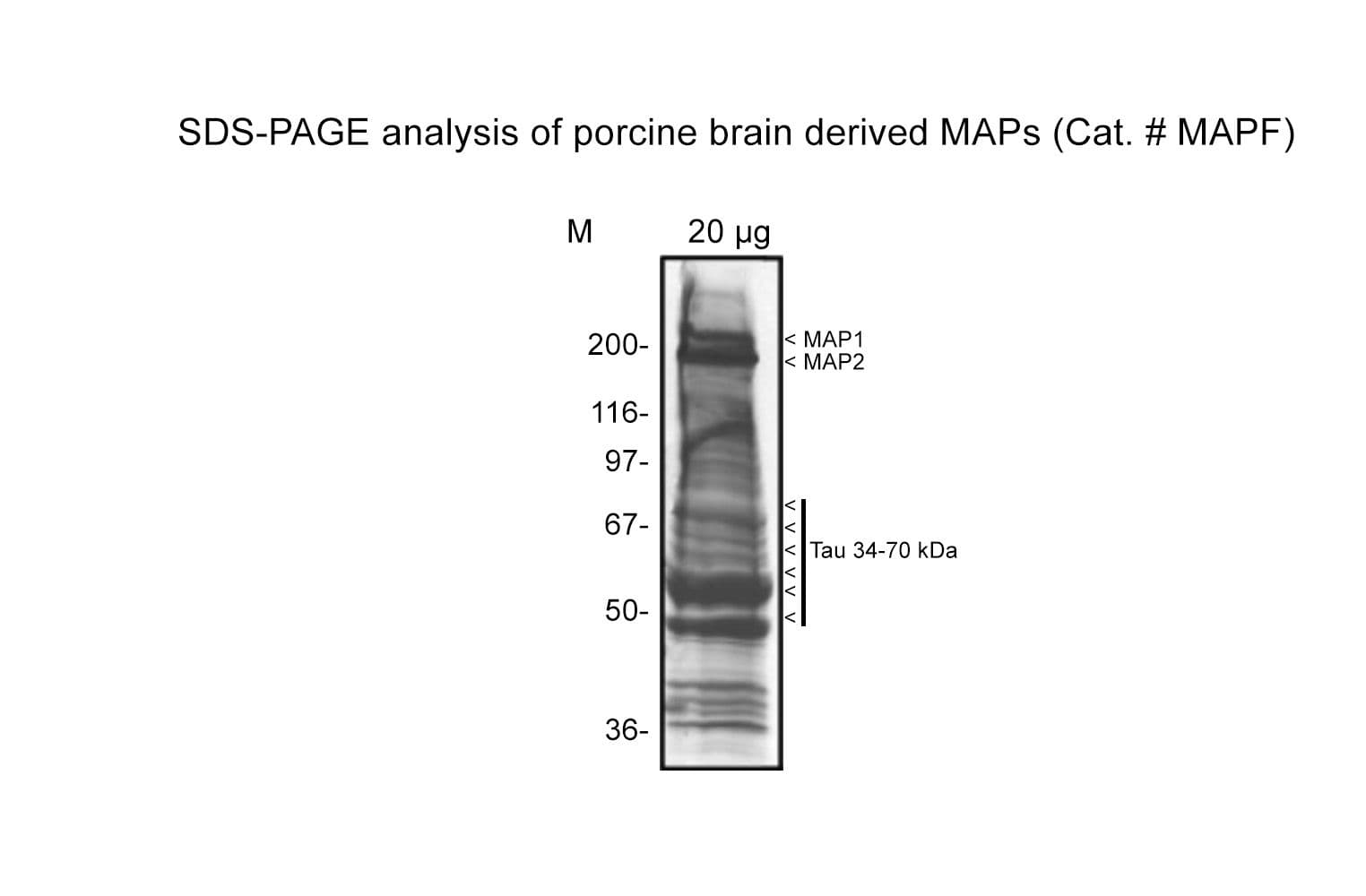
A native microtubule-associated protein (MAP) fraction has been isolated from bovine brain by temperature-induced tubulin polymerization and ionic exchange chromatography.
- Molecular weight: broad range of enriched MAPs, major bands at ~250 kDa (MAP 1 & 2) and ~37-50 kDa (Tau isotypes)
- Formulation: supplied as a lyophilized powder
- Composition: when resuspended to 1 mg/ml the buffer composition is; 10 mM PIPES pH 7.5, 0.3 mM EGTA, and 3% (w/v) sucrose