Tubulin polymerization HTS assay using >97% pure tubulin, OD based - Porcine (BK004P)
Product Uses Include
- Screening compounds for effects on tubulin polymerization activity.
- Screening proteins for effects on tubulin polymerization activity.
- Teaching aid for undergraduate/graduate class in pharmacology.
- Economical approach to HTS with tubulin.
Introduction
This assay is based on an adaptation of the original method of Shelanski et al. and Lee et al. (1,2), which demonstrated that light is scattered by microtubules to an extent that is proportional to the concentration of microtubule polymer. The resulting polymerization curve is representative of the three phases of microtubule polymerization, namely nucleation, growth and steady state equilibrium. See the About Tubulin page for more information. The assay is optimized for a 96-well format for low CVs and large sample number handling.
This kit contains "HTS tubulin", which is >97% pure (Cat. # HTS03). The same assay is also available with our highest purity tubulin (>99% pure, Cat. # T240) and is sold under Cat. # BK006P. BK004P provides an economical alternative to the higher purity BK006P assay and can be a good choice for large primary screens. Cytoskeleton, Inc. also provides a fluorescence based tubulin polymerization assay in miniaturized format (Cat. # BK011P), which is also ideal for high throughput screening.
If you are interested in using either of these tubulin polymerization assays in a high throughput setting, please contact our technical service department for advice and bulk quotes.
Kit contents
This kit contains enough materials for 24 assays (BK004P). The following reagents are included:
- HTS tubulin, lyophilized (Cat. # HTS03)
- GTP solution (Cat. # BST06)
- General tubulin buffer (PEM, Cat. # BST01)
- Tubulin glycerol buffer (Cat. # BST05)
- Paclitaxel positive control (Cat. # TXD01)
- DMSO for paclitaxel.
- Half area 96-well plate for polymerization reactions
- Manual with detailed protocols and extensive troubleshooting guide.
Equipment needed
- 96-well plate spectrophotometer with filters to read optical density at 340 nm.
- Multi-channel pipette for rapid pipetting of tubulin
Example results
The BK004P kit was used to study the effect of 48 different conditions on tubulin polymerization rates in a 96 well format. Each condition was tested in duplicate samples (Fig. 1)
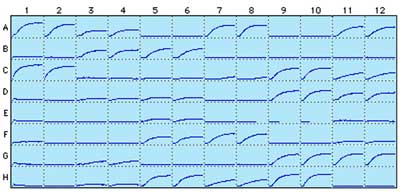
Figure 1. Tubulin polymerization in 96-well format using BK004P. 48 different tubulin polymerization conditions were tested in duplicates. Wells A1 and A2 represent a control reaction and wells C1 and C2 represent the addition of the microtubule stabilizing drug Paclitaxel, resulting in an increased polymerization rate. Each well represents one polymerization reaction. The x-axis is 20 min and the y-axis corresponds to 0.0-0.2 OD at 340 nm.
References
- Shelanski, M. L., Gaskin, F. and Cantor, C. R. (1973). Microtubule assembly in the absence of added nucleotides. Proc. Natl. Acad. Sci. U.S.A. 70, 765-768.
- Lee, J. C. and Timasheff, S. N. (1977). In vitro reconstitution of calf brain microtubules: effects of solution variable. Biochemistry, 16, 1754-1762.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
 For our Tubulin Polymerization (Absorbance) Excel Template please download here.
For our Tubulin Polymerization (Absorbance) Excel Template please download here. For our IC50 from Vmax Polymerization Excel Template please download here.
For our IC50 from Vmax Polymerization Excel Template please download here.
Question 1: What is the difference between this polymerization kit and BK006P?
Answer 1: Both the BK004P and BK006P are tubulin polymerization kits that are absorbance-based rather than fluorescence-based. The only difference between the two absorbance-based kits is that BK004P uses 97% pure tubulin (remaining 3% are MAPs) while BK006P uses >99% pure tubulin. This is an important difference because the presence of MAPs means that tubulin polymerization can be examined in the absence of enhancers such as glycerol or taxol with as little as 3 or 4 mg/ml tubulin using the BK004P kit. In this case MAPs act as polymerization enhancers. With BK006P, an enhancer such as glycerol or taxol must be used to drive tubulin polymerization with concentrations <5 mg/ml tubulin. Using tubulin at 5 mg/ml or higher allows for the omission of glycerol or taxol. In some cases, glycerol can interfere with the binding of tubulin accessory proteins or compounds/reagents/drugs. Assay conditions can easily be altered to test for glycerol interference.
Question 2: Which kit is best for screening a compound/reagent/drµg for its effects on tubulin polymerization?
Answer 2: All 3 tubulin polymerization kits (2 absorbance-based kits, BK004P and BK006P; 1 fluorescence-based kit, BK011P) are well-suited for screening of potential tubulin polymerization enhancers and inhibitors. Each kit has its own pros and cons. For initial compound/drµg screening, we recommend the absorbance-based tubulin polymerization assay BK004P because it’s economical. This kit uses 97% pure tubulin (remaining 3% are MAPs) while BK006P and BK011P use >99% pure tubulin. This is an important difference because the presence of MAPs means that tubulin polymerization can be examined in the absence of enhancers or inhibitors with as little as 3 or 4 mg/ml tubulin using the BK004P kit. To study enhancers, we recommend using 3 mg/ml tubulin, whereas 4 mg/ml tubulin is recommended for inhibitors. In the case of BK004P, MAPs act as polymerization enhancers. With BK006P and BK011P, an enhancer such as glycerol or taxol must be used to drive tubulin polymerization with concentrations <5 mg/ml tubulin. Using tubulin at 5 mg/ml or higher allows for the omission of glycerol or taxol, but requires additional tubulin. In some cases, glycerol can interfere with the binding of tubulin accessory proteins or compounds. However, since BK011P is fluorescence-based, there is increased sensitivity that allows the researcher to use 1/3 as much tubulin with greater sensitivity. Thus, the kit provides 96 assays versus the 30 assays of BK004P or BK006P, thus BK011P is the most economical when requiring >30 assays for the project.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com




