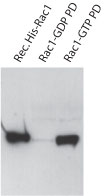
| Author | Title | Journal | Year | Article Link |
|---|
| Zhang, Huimin et al. | PREX1 improves homeostatic proliferation to maintain a naive CD4+ T cell compartment in older age | JCI Insight | 2024 | ISSN 0021--9738 |
| Veloso, Alexandra et al. | The cytoskeleton adaptor protein Sorbs1 controls the development of lymphatic and venous vessels in zebrafish | BMC Biology 2024 22:1 | 2024 | ISSN 1741--7007 |
| Bahcheli, Alexander T. et al. | Pan-cancer ion transport signature reveals functional regulators of glioblastoma aggression | The EMBO journal | 2024 | ISSN 1460-2075 |
| Chen, Weiwei et al. | Tumour-associated macrophage-derived DOCK7-enriched extracellular vesicles drive tumour metastasis in colorectal cancer via the RAC1/ABCA1 axis | Clinical and Translational Medicine | 2024 | ISSN 2001--1326 |
| Aljagthmi, Amjad A. et al. | ∆Np63α inhibits Rac1 activation and cancer cell invasion through suppression of PREX1 | Cell Death Discovery 2024 10:1 | 2024 | ISSN 2058--7716 |
| Koo, Tin Yan et al. | N-Acryloylindole-alkyne (NAIA) enables imaging and profiling new ligandable cysteines and oxidized thiols by chemoproteomics | Nature Communications 2023 14:1 | 2023 | ISSN 2041--1723 |
| Newman, Daniel et al. | 3D matrix adhesion feedback controls nuclear force coupling to drive invasive cell migration | Cell reports | 2023 | ISSN 2211--1247 |
| Chen, Di et al. | Knockdown of Porf-2 restores visual function after optic nerve crush injury | Cell Death & Disease 2023 14:8 | 2023 | ISSN 2041--4889 |
| Yu, Shuxiang et al. | ELMO1 Deficiency Reduces Neutrophil Chemotaxis in Murine Peritonitis | International Journal of Molecular Sciences | 2023 | ISSN 1422-0067 |
| Yang, Yuchen et al. | β-hydroxybutyrate impairs the directionality of migrating neutrophils through inhibiting the autophagy-dependent degradation of Cdc42 and Rac1 in ketotic cows | Journal of Dairy Science | 2023 | |
| Serwe, Guillaume et al. | CNK2 promotes cancer cell motility by mediating ARF6 activation downstream of AXL signalling | Nature Communications 2023 14:1 | 2023 | ISSN 2041--1723 |
| Yau, Eric et al. | SP-R210 isoforms of Myosin18A modulate endosomal sorting and recognition of influenza A virus infection in macrophages | Microbes and Infection | 2023 | ISSN 1286--4579 |
| Wang, Yueyang et al. | Atypical peripheral actin band formation via overactivation of RhoA and nonmuscle myosin II in mitofusin 2-deficient cells | eLife | 2023 | ISSN 2050-084X |
| Morretton, Jean-Philippe et al. | A catalog of numerical centrosome defects in epithelial ovarian cancers | EMBO Molecular Medicine | 2022 | ISSN 1757--4684 |
| Karthikeyan, Subbulakshmi et al. | RAB4A GTPase regulates epithelial-to-mesenchymal transition by modulating RAC1 activation | Breast Cancer Research | 2022 | ISSN 1465-542X |
| Singh, Neeraj et al. | BACE-1 inhibition facilitates the transition from homeostatic microglia to DAM-1 | Science Advances | 2022 | ISSN 2375-2548 |
| Wang, Yuting et al. | EPS8L1 promotes migration and metastasis of ovarian cancer by activating Rac1/MAPK signaling pathway via upregulating TIAM2 | Authorea Preprints | 2022 | Article Link |
| Adan, Hanad et al. | Activated Src requires Cadherin-11, Rac, and gp130 for Stat3 activation and survival of mouse Balb/c3T3 fibroblasts | Cancer Gene Therapy 2022 29:10 | 2022 | ISSN 1476--5500 |
| Usuki, Seigo et al. | Konjac Ceramide (kCer)-Mediated Signal Transduction of the Sema3A Pathway Promotes HaCaT Keratinocyte Differentiation | Biology | 2022 | ISSN 2079-7737 |
| Adan, H et al. | Activated Src requires Cadherin-11, Rac, and gp130 for Stat3 activation and survival of mouse Balb/c3T3 fibroblasts | Cancer Gene … | 2022 | Article Link |
| Khan, Alamzeb et al. | ArhGEF12 activates Rap1A and not RhoA in human dermal microvascular endothelial cells to reduce tumor necrosis factor-induced leak | FASEB journal : official publication of the Federation of American Societies for Experimental Biology | 2022 | ISSN 1530-6860 |
| Agbaegbu Iweka, Chinyere et al. | The lipid phosphatase-like protein PLPPR1 associates with RhoGDI1 to modulate RhoA activation in response to axon growth inhibitory molecules | Journal of Neurochemistry | 2021 | ISSN 1471-4159 |
| Masi, Ilenia et al. | Endothelin-1 drives invadopodia and interaction with mesothelial cells through ILK | Cell Reports | 2021 | ISSN 2211-1247 |
| Bai, Xiaoyuan et al. | Induction of cyclophilin A by influenza A virus infection facilitates group A Streptococcus coinfection | Cell Reports | 2021 | ISSN 2211-1247 |
| Wang, Jiang Lin et al. | Spinophilin modulates pain through suppressing dendritic spine morphogenesis via negative control of Rac1-ERK signaling in rat spinal dorsal horn | Neurobiology of Disease | 2021 | ISSN 1095-953X |
| Guo, Shuyu et al. | Trio cooperates with Myh9 to regulate neural crest-derived craniofacial development | Theranostics | 2021 | ISSN 1838-7640 |
| Wang, Junyi et al. | Rho-GEF Trio regulates osteosarcoma progression and osteogenic differentiation through Rac1 and RhoA | Cell Death and Disease | 2021 | ISSN 2041-4889 |
| Zhou, Wenqing et al. | Mitofusin 2 regulates neutrophil adhesive migration and the actin cytoskeleton | Journal of Cell Science | 2021 | ISSN 1477-9137 |
| Li, Chunsen et al. | GEFT Inhibits Autophagy and Apoptosis in Rhabdomyosarcoma via Activation of the Rac1/Cdc42-mTOR Signaling Pathway | Frontiers in Oncology | 2021 | ISSN 2234-943X |
| McCray, Brett A. et al. | Neuropathy-causing TRPV4 mutations disrupt TRPV4-RhoA interactions and impair neurite extension | Nature Communications | 2021 | ISSN 2041-1723 |
| Shin, Seung Kak et al. | Exogenous 8-hydroxydeoxyguanosine ameliorates liver fibrosis through the inhibition of Rac1-NADPH oxidase signaling | Journal of gastroenterology and hepatology | 2020 | ISSN 1440--1746 |
| Benz, Peter M. et al. | AKAP12 deficiency impairs VEGF-induced endothelial cell migration and sprouting | Acta physiologica (Oxford, England) | 2020 | ISSN 1748--1716 |
| Thamilselvan, Vijayalakshmi et al. | P-Rex1 Mediates Glucose-Stimulated Rac1 Activation and Insulin Secretion in Pancreatic β-Cells | Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology | 2020 | ISSN 1421--9778 |
| Li, Zean et al. | The metastatic promoter DEPDC1B induces epithelial‐mesenchymal transition and promotes prostate cancer cell proliferation via Rac1‐PAK1 signaling | Clinical and Translational Medicine | 2020 | ISSN 2001--1326 |
| Moody, Jasmine C. et al. | The Rho-GEF PIX-1 directs assembly or stability of lateral attachment structures between muscle cells | Nature Communications | 2020 | ISSN 2041-1723 |
| Danelon, Victor et al. | Modular and Distinct Plexin-A4/FARP2/Rac1 Signaling Controls Dendrite Morphogenesis | Journal of Neuroscience | 2020 | ISSN 1529-2401 |
| Larribère, Lionel et al. | NF1-RAC1 axis regulates migration of the melanocytic lineage | Translational Oncology | 2020 | ISSN 1936-5233 |
| Ichikawa, Takehiko et al. | Non-junctional role of Cadherin3 in cell migration and contact inhibition of locomotion via domain-dependent, opposing regulation of Rac1 | Scientific Reports | 2020 | ISSN 2045-2322 |
| Wei, Yiju et al. | NEDD 4L‐mediated Merlin ubiquitination facilitates Hippo pathway activation | EMBO reports | 2020 | ISSN 1469--221X |
| Aladowicz, Ewa et al. | Shcd binds dock4, promotes ameboid motility and metastasis dissemination, predicting poor prognosis in melanoma | Cancers | 2020 | ISSN 2072-6694 |
| Wang, Ruixiao et al. | Rac1 silencing, NSC23766 and EHT1864 reduce growth and actin organization of bladder smooth muscle cells | Life Sciences | 2020 | ISSN 1879-0631 |
| Smalley, Tracess et al. | The Atypical Protein Kinase C Small Molecule Inhibitor ζ-Stat, and Its Effects on Invasion Through Decreases in PKC-ζ Protein Expression | Frontiers in Oncology | 2020 | ISSN 2234-943X |
| Barabutis, Nektarios et al. | Protective mechanism of the selective vasopressin V1A receptor agonist selepressin against endothelial barrier dysfunction | Journal of Pharmacology and Experimental Therapeutics | 2020 | ISSN 1521-0103 |
| Joshi, Rakesh et al. | DLC1 SAM domain-binding peptides inhibit cancer cell growth and migration by inactivating RhoA | Journal of Biological Chemistry | 2020 | ISSN 1083-351X |
| Chen, Lixia et al. | CSRP2 suppresses colorectal cancer progression via p130Cas/Rac1 axis-meditated ERK, PAK, and HIPPO signaling pathways | Theranostics | 2020 | ISSN 1838-7640 |
| Lian, Eric Y. et al. | RET isoforms contribute differentially to invasive processes in pancreatic ductal adenocarcinoma | Oncogene | 2020 | ISSN 1476-5594 |
| Amato, Clelia et al. | WASP Restricts Active Rac to Maintain Cells’ Front-Rear Polarization | Current Biology | 2019 | ISSN 0960-9822 |
| Tsygankova, Oxana M. et al. | A unique role for clathrin light chain A in cell spreading and migration | Journal of Cell Science | 2019 | ISSN 1477-9137 |
| Park, Jin Seok et al. | Switch-like enhancement of epithelial-mesenchymal transition by YAP through feedback regulation of WT1 and Rho-family GTPases | Nature Communications | 2019 | ISSN 2041-1723 |
| Lang, Yue et al. | MiR-30 family prevents uPAR-ITGB3 signaling activation through calcineurin-NFATC pathway to protect podocytes | Cell Death and Disease | 2019 | ISSN 2041-4889 |
| Dagliyan, Onur et al. | Engineering proteins for allosteric control by light or ligands | Nature Protocols | 2019 | ISSN 1750-2799 |
| Zhou, Yi Fan et al. | Sema3E/PlexinD1 signaling inhibits postischemic angiogenesis by regulating endothelial DLL4 and filopodia formation in a rat model of ischemic stroke | FASEB Journal | 2019 | ISSN 1530-6860 |
| Liu, Chunxia et al. | Epigenetically upregulated GEFT-derived invasion and metastasis of rhabdomyosarcoma via epithelial mesenchymal transition promoted by the Rac1/Cdc42-PAK signalling pathway | EBioMedicine | 2019 | ISSN 2352-3964 |
| Liu, Yunlong et al. | Social Isolation Induces Rac1-Dependent Forgetting of Social Memory | Cell Reports | 2018 | ISSN 2211-1247 |
| McQueeney, Kelley E. et al. | Targeting ovarian cancer and endothelium with an allosteric PTP4A3 phosphatase inhibitor | Oncotarget | 2018 | ISSN 1949-2553 |
| Zago, Giulia et al. | Ralb directly triggers invasion downstream ras by mobilizing the wave complex | eLife | 2018 | ISSN 2050-084X |
| Barabutis, Nektarios et al. | Wild-type p53 enhances endothelial barrier function by mediating RAC1 signalling and RhoA inhibition | Journal of Cellular and Molecular Medicine | 2018 | ISSN 1582-1838 |
| Shi, Hao et al. | Hippo Kinases Mst1 and Mst2 Sense and Amplify IL-2R-STAT5 Signaling in Regulatory T Cells to Establish Stable Regulatory Activity | Immunity | 2018 | ISSN 1097-4180 |
| Hayashi, Kentaro et al. | Intracellular calcium signal at the leading edge regulates mesodermal sheet migration during Xenopus gastrulation | Scientific Reports | 2018 | ISSN 2045-2322 |
| Shang, Wanjing et al. | Genome-wide CRISPR screen identifies FAM49B as a key regulator of actin dynamics and T cell activation | Proceedings of the National Academy of Sciences of the United States of America | 2018 | ISSN 1091-6490 |
| Yang, Huan et al. | Cytotoxic Necrotizing Factor 1 Downregulates CD36 Transcription in Macrophages to Induce Inflammation During Acute Urinary Tract Infections | Frontiers in Immunology | 2018 | ISSN 1664-3224 |
| Yao, Zhihui et al. | P311 Accelerates Skin Wound Reepithelialization by Promoting Epidermal Stem Cell Migration Through RhoA and Rac1 Activation | Stem Cells and Development | 2017 | ISSN 1557-8534 |
| Guo, Yaxiu et al. | Cytotoxic necrotizing factor 1 promotes prostate cancer progression through activating the Cdc42–PAK1 axis | Journal of Pathology | 2017 | ISSN 1096-9896 |
| Wang, Yan et al. | Involvement of Rac1 signalling pathway in the development and maintenance of acute inflammatory pain induced by bee venom injection | British Journal of Pharmacology | 2016 | ISSN 1476-5381 |
| Han, Jie et al. | Farnesyl pyrophosphate synthase inhibitor, ibandronate, improves endothelial function in spontaneously hypertensive rats | Molecular Medicine Reports | 2016 | ISSN 1791-3004 |
| Zhan, Rixing et al. | Nitric oxide promotes epidermal stem cell migration via cGMP-Rho GTPase signalling | Scientific Reports | 2016 | ISSN 2045-2322 |
| Tie, S. R. et al. | Regulation of sarcoma cell migration, invasion and invadopodia formation by AFAP1L1 through a phosphotyrosine-dependent pathway | Oncogene | 2016 | ISSN 1476-5594 |
| Wang, Y. et al. | Inhibition of prostate smooth muscle contraction and prostate stromal cell growth by the inhibitors of Rac, NSC23766 and EHT1864 | British Journal of Pharmacology | 2015 | ISSN 1476-5381 |
| Wang, Shi Jie et al. | CD147 promotes Src-dependent activation of Rac1 signaling through STAT3/DOCK8 during the motility of hepatocellular carcinoma cells | Oncotarget | 2015 | ISSN 1949-2553 |
| Zhan, Rixing et al. | Nitric oxide enhances keratinocyte cell migration by regulating Rho GTPase via cGMP-PKG signalling | PLoS ONE | 2015 | ISSN 1932-6203 |
| Tang, Xiaoyun et al. | Lipid phosphate phosphatase-1 expression in cancer cells attenuates tumor growth and metastasis in mice | Journal of Lipid Research | 2014 | ISSN 1539-7262 |
| Hara, Yusuke et al. | Directional migration of leading-edge mesoderm generates physical forces: Implication in Xenopus notochord formation during gastrulation | Developmental Biology | 2013 | ISSN 1095-564X |
| Han, Gangwen et al. | Preventive and therapeutic effects of Smad7 on radiation-induced oral mucositis | Nature Medicine | 2013 | ISSN 1078-8956 |
| Geletu, M. et al. | Classical cadherins control survival through the gp130/Stat3 axis | Biochimica et Biophysica Acta - Molecular Cell Research | 2013 | ISSN 0167-4889 |
| Zallocchi, Marisa et al. | α1β1 Integrin/rac1-dependent mesangial invasion of glomerular capillaries in alport syndrome | American Journal of Pathology | 2013 | ISSN 0002-9440 |
| Nithipatikom, Kasem et al. | Cannabinoid receptor type 1 (CB1) activation inhibits small GTPase RhoA activity and regulates motility of prostate carcinoma cells | Endocrinology | 2012 | ISSN 1945--7170 |
| Lee, Wonhwa et al. | Barrier protective effects of withaferin A in HMGB1-induced inflammatory responses in both cellular and animal models | Toxicology and applied pharmacology | 2012 | ISSN 1096--0333 |
| Ninkovi, Jana et al. | Morphine decreases bacterial phagocytosis by inhibiting actin polymerization through cAMP-, Rac-1-, and p38 MAPK-dependent mechanisms | American Journal of Pathology | 2012 | ISSN 0002-9440 |
| Wong, Hon Kit et al. | Merlin/NF2 regulates angiogenesis in schwannomas through a Rac1/semaphorin 3F-dependent mechanism | Neoplasia | 2012 | ISSN 1476-5586 |
| Gastonguay, Adam et al. | The role of Rac1 in the regulation of NF-kB activity, cell proliferation, and cell migration in non-small cell lung carcinoma | Cancer Biology & Therapy | 2012 | ISSN 1538--4047 |
| Syed, Ismail et al. | L-threo-C 6 -pyridinium-ceramide bromide, a novel cationic ceramide, induces NADPH oxidase activation, mitochondrial dysfunction and loss in cell viability in INS 832/13 β-cells | Cellular Physiology and Biochemistry | 2012 | ISSN 1421-9778 |
| Ock, Chan Young et al. | A novel approach for stress-induced gastritis based on paradoxical anti-oxidative and anti-inflammatory action of exogenous 8-hydroxydeoxyguanosine | Biochemical pharmacology | 2011 | ISSN 1873--2968 |
| Garkavtsev, Igor et al. | Dehydro-α-lapachone, a plant product with antivascular activity | Proceedings of the National Academy of Sciences of the United States of America | 2011 | ISSN 0027-8424 |
| Souma, Tomokazu et al. | Luminal alkalinization attenuates proteinuria-induced oxidative damage in proximal tubular cells | Journal of the American Society of Nephrology | 2011 | ISSN 1046-6673 |
| Skalski, Michael et al. | SNARE-mediated membrane traffic is required for focal adhesion kinase signaling and Src-regulated focal adhesion turnover | Biochimica et Biophysica Acta - Molecular Cell Research | 2011 | ISSN 0167-4889 |
| Vidal-Quadras, Maite et al. | Rac1 and Calmodulin Interactions Modulate Dynamics of ARF6-Dependent Endocytosis | Traffic | 2011 | ISSN 1398-9219 |
| Balanis, Nikolas et al. | β3 integrin-EGF receptor cross-talk activates p190RhoGAP in mouse mammary gland epithelial cells | Molecular Biology of the Cell | 2011 | ISSN 1059-1524 |
| Jayaram, Bhavaani et al. | Isoprenylcysteine carboxyl methyltransferase facilitates glucose-induced Rac1 activation, ROS generation and insulin secretion in INS 832/13 β-cells | Islets | 2011 | ISSN 1938-2014 |
| McAuley, Erin M. et al. | Phenylboronic acid is a more potent inhibitor than boric acid of key signaling networks involved in cancer cell migration | Cell Adhesion and Migration | 2011 | ISSN 1933-6926 |
| Kobayashi, Takashi et al. | Activation of Rac1 is closely related to androgen-independent cell proliferation of prostate cancer cells both in vitro and in vivo | Molecular Endocrinology | 2010 | ISSN 0888-8809 |
| Medhora, Meetha et al. | 20-HETE increases superoxide production and activates NAPDH oxidase in pulmonary artery endothelial cells | American Journal of Physiology - Lung Cellular and Molecular Physiology | 2008 | ISSN 1040-0605 |
| Bergstrom, R. A. et al. | Agrin induced morphological and structural changes in growth cones of cultured hippocampal neurons | Neuroscience | 2007 | ISSN 0306-4522 |
| Wang, Guoyan et al. | Genetic ablation of Rac1 in cartilage results in chondrodysplasia | Developmental Biology | 2007 | ISSN 0012-1606 |
| Slice, Lee W. et al. | Angiotensin II and epidermal growth factor induce cyclooxygenase-2 expression in intestinal epithelial cells through small GTPases using distinct signaling pathways | The Journal of biological chemistry | 2005 | ISSN 0021--9258 |
| Sasai, Noriaki et al. | The neurotrophin-receptor-related protein NRH1 is essential for convergent extension movements | | 2004 | PMID 15258592 |
| Yang, Seun Ah et al. | Rho and Rho-kinase mediate thrombin-induced phosphatidylinositol 4-phosphate 5-kinase trafficking in platelets | Journal of Biological Chemistry | 2004 | ISSN 0021-9258 |
| Zhang, Yongjun et al. | Distinct roles of two structurally closely related focal adhesion proteins, alpha-parvins and beta-parvins, in regulation of cell morphology and survival | The Journal of biological chemistry | 2004 | ISSN 0021--9258 |
| Vogl, Thomas et al. | MRP8 and MRP14 control microtubule reorganization during transendothelial migration of phagocytes | Blood | 2004 | ISSN 0006-4971 |
| Chromy, Brett A. et al. | Self-assembly of Abeta(1-42) into globular neurotoxins | Biochemistry | 2003 | ISSN 0006--2960 |
| Quadri, Sadiqa K. et al. | Endothelial Barrier Strengthening by Activation of Focal Adhesion Kinase * | Journal of Biological Chemistry | 2003 | ISSN 0021--9258 |