Alpha-actinin protein: rabbit skeletal muscle
Cat. #AT01
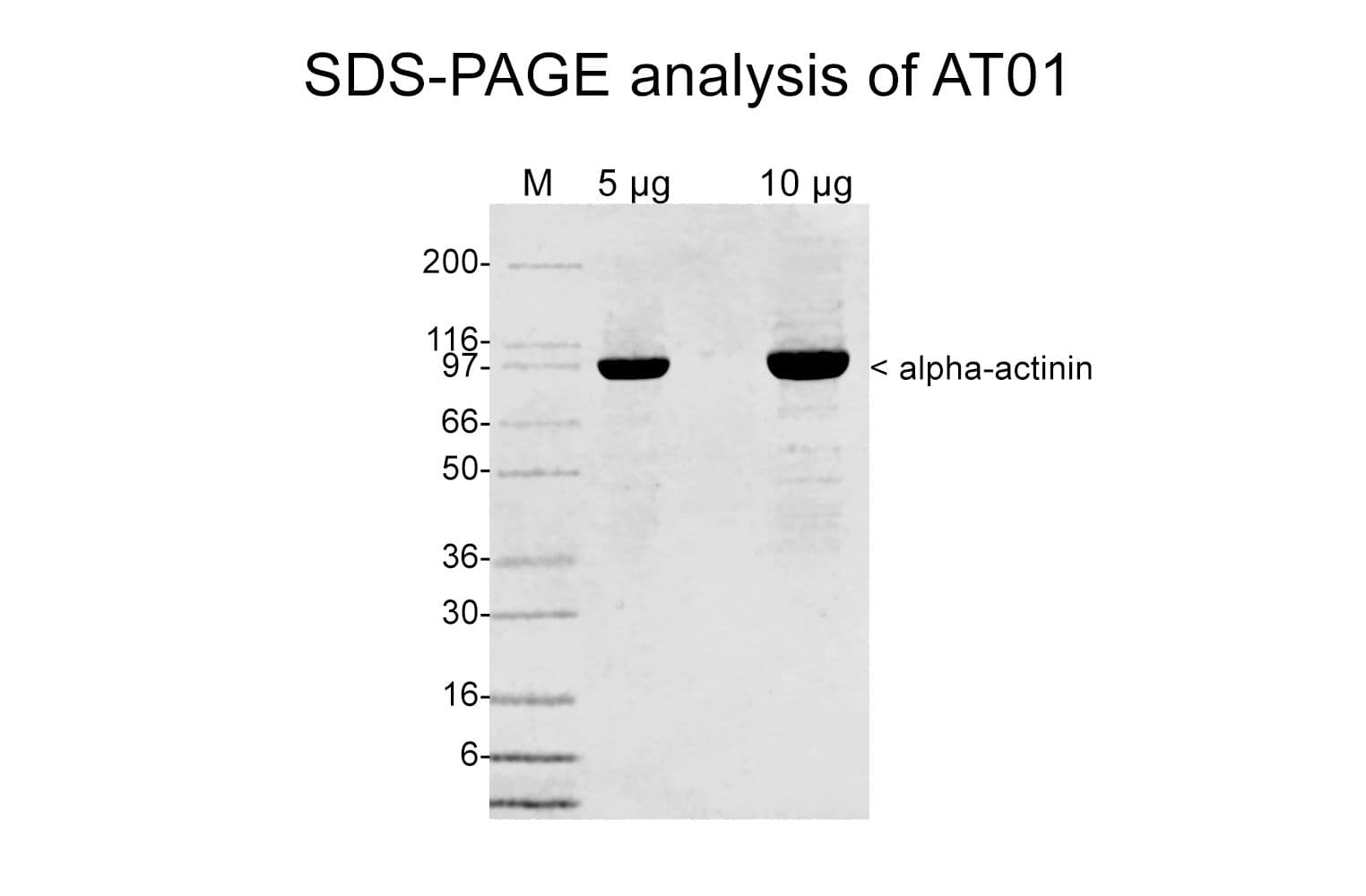


Cat. #AT01



Alpha-actinin is a cytoskeletal actin-binding protein that cross-links actin filaments and anchors them to various cellular structures, contributing to cell shape, motility, and adhesion. It plays critical roles in muscle sarcomere organization and non-muscle cellular architecture, making it essential for structural integrity and signaling functions.
Alpha-actinin is purified from rabbit skeletal muscle
Protein purity is determined by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity is >85%
The biological activity of alpha-actinin is assessed by its ability to bind to F-actin filaments. An F-actin binding assay is performed. Under the assay conditions described below,>65% of alpha-actinin co-pellets with F-actin, while <10% pellets in the absence of F-actin (see datasheet for details)