Actin Polymerization Biochem Kit (fluorescence format): rabbit skeletal muscle actin
Product Uses Include
- To show quantitative / qualitative effects on actin polymerization by the addition of a extract, an actin binding protein or compound.
- To show quantitative / qualitative effects on actin polymerization by addition of an F-actin nucleating protein, compound or extract.
- To show quantitative / qualitative effects on steady-state F-actin levels by addition of an F-actin severing protein, compound or extract.
- To show quantitative / qualitative effects on actin depolymerization by addition of an actin binding protein, compound or extract.
Introduction
The Actin Polymerization Biochem Kit™ is based on the enhanced fluorescence of pyrene conjugated actin that occurs during polymerization. The enhanced fluorescence that occurs when pyrene G-actin (monomer) forms pyrene F-actin can be measured in a fluorimeter to follow polymerization over time. Also, by using preformed pyrene F-actin, it is possible to follow depolymerization. Both cell/tissue extracts and purified proteins can be added to the reaction mixture to identify their effect on actin polymerization. The components of the kit can also be used separately for other actin based assays such as a spin-down assays to detect F-actin binding proteins (see also BK001) or size exclusion chromatography to identify G-actin binding proteins. See the About Actin page for more information on assays testing actin binding proteins.
While this kit comes with pyrene labeled skeletal muscle actin, it can also be used to study polymerization of other types of actin such as non-muscle actin (Cat. # APHL99) or cardiac actin (Cat. # AD99). Polymerization assays with these actins can be performed using a 10:1 ratio between the actin you want to study and the included pyrene actin
Kit contents
The kit contains enough materials for 30-100 assays depending on assay volume. The following reagents are included:
- 5 x 1 mg Pyrene labeled actin (Cat. # AP05).
- General Actin Buffer (Cat. # BSA01).
- Actin Polymerization Buffer (Cat. # BSA02).
- ATP 100mM (Cat. # BSA04).
- Tris-HCl pH 7.5, 100 mM
- Manual with detailed protocols and extensive troubleshooting guide.
Equipment needed
- Fluorescence spectrophotometer (cuvette or 96-well plate) with 4-10 nm bandwidth at 365 nm excitation wavelength, and 4-10 nm bandwidth at 407 nm emission wavelength.
- Small capacity (100-1000 µl) fluorescence spectrophotometer cuvette or 96-well plate.
Example results
The Actin Polymerization Biochem Kit™ was used to study the effects of Arp2/3 (Cat. # RP01) and the VCA domain of WASP (Cat. # VCG03) on actin polymerization rates. The Arp2/3 complex is an actin filament nucleator but has low nucleating/polymerizing activity on its own. The VCA domain of WASP is an activator of the Arp2/3 complex. Hence, when the Arp2/3 complex is mixed with the WASP VCA domain, these two exert a potent actin polymerizing activity (Fig. 1).
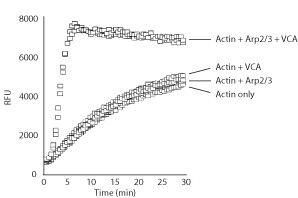
Figure 1. Actin polymerization stimulated by Arp2/3 complex and the VCA domain of WASP. Actin polymerization was measured using kit BK003. The addition of Arp2/3 complex or the VCA domain alone to actin has minimal effects on actin polymerization, while the combination of Arp2/3 and the VCA domain strongly stimulates the rate of actin polymerization.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
![]() For our Actin Polymerization Excel Template please download here.
For our Actin Polymerization Excel Template please download here.
Question 1: Can I use this polymerization kit to measure polymerization of non-muscle actin?
Answer 1: Pyrene-labeled non-muscle actin has been shown to be unstable under normal storage conditions. To examine the polymerization of unlabeled non-muscle actin, please click here for a polymerization protocol that uses an excess of unlabeled non-muscle, cardiac or gizzard actin (Cat# APHL95, APHL99, AS99) + a small amount of pyrene-labeled skeletal muscle actin (Cat. # AP05). The pyrene muscle actin will not polymerize efficiently on its own at the concentration used in this assay, so the reaction is dependent on unlabeled actin polymerization for F-actin formation. In this way, the pyrene-labeled muscle actin is taken up and polymerized to serve as a reporter for polymerization of the unlabeled actin that is present at a much greater concentration.
Question 2: Can cell lysates be used with this kit as the source of a test protein?
Answer 2: Yes, cell lysates can be used as the source of the test protein for examining actin polymerization enhancers and inhibitors. However, Cytoskeleton does not recommend this as the purity and concentration of the protein will often be too low to interact with actin. Also, the lysates will contain additional accessory proteins and multiple phosphatases and proteases that can interfere or alter the interactions between actin and test protein. If lysates are to be used, we recommend the following:
Although this kit is designed for use with pure proteins or compounds, some researchers have added extracts with good results. Generally researchers use over-expressed proteins and a wild-type control extract similarly over-expressed. It is necessary to make a 10 mg/ml protein extract and then use 1/3rd volume of this to 2/3rd volume of pyrene-actin (Cat. # AP05). In this way there is a high enough concentration of protein to make a difference. The extraction buffer should be 20 mM Hepes pH 7.5, 20 mM NaCl, plus any co-factors for your protein, and a protease inhibitor cocktail (e.g., Cat# PIC02). Rinse the cells with an ice cold buffer and lyse cells with a 25 g bent over syringe needle or other device. The control cell line is very critical because the actin polymerization reaction is very sensitive to slight differences in protein concentration or salts.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com









