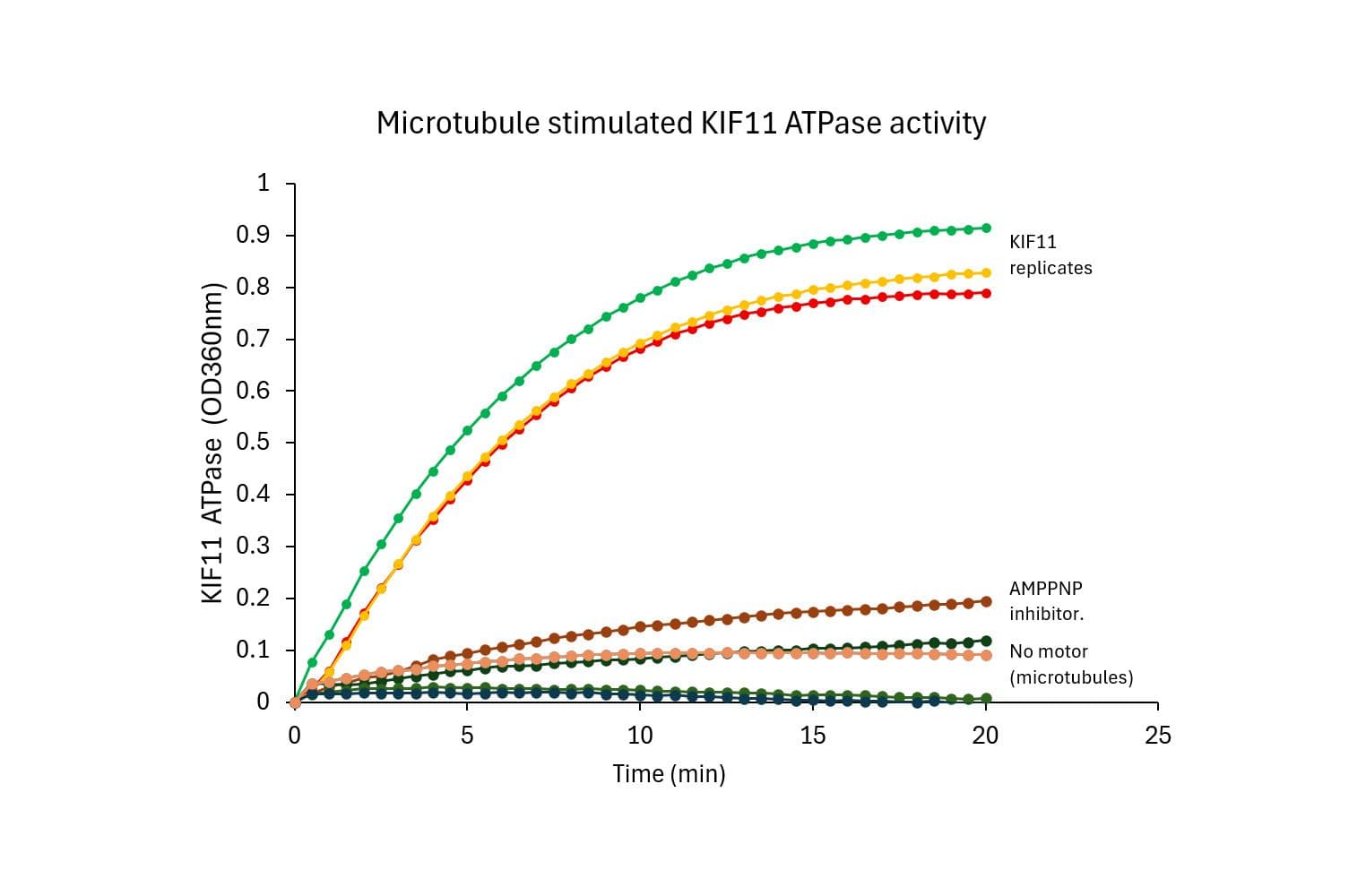
KIF11—also known as kinesin-5 or Eg5—is an essential mitotic motor protein that drives spindle pole separation and bipolar spindle assembly. Several small molecule inhibitors targeting KIF11 have been explored as anti-mitotic cancer therapies, though clinical efficacy as monotherapies has been limited, with filanesib showing promise in multiple myeloma.
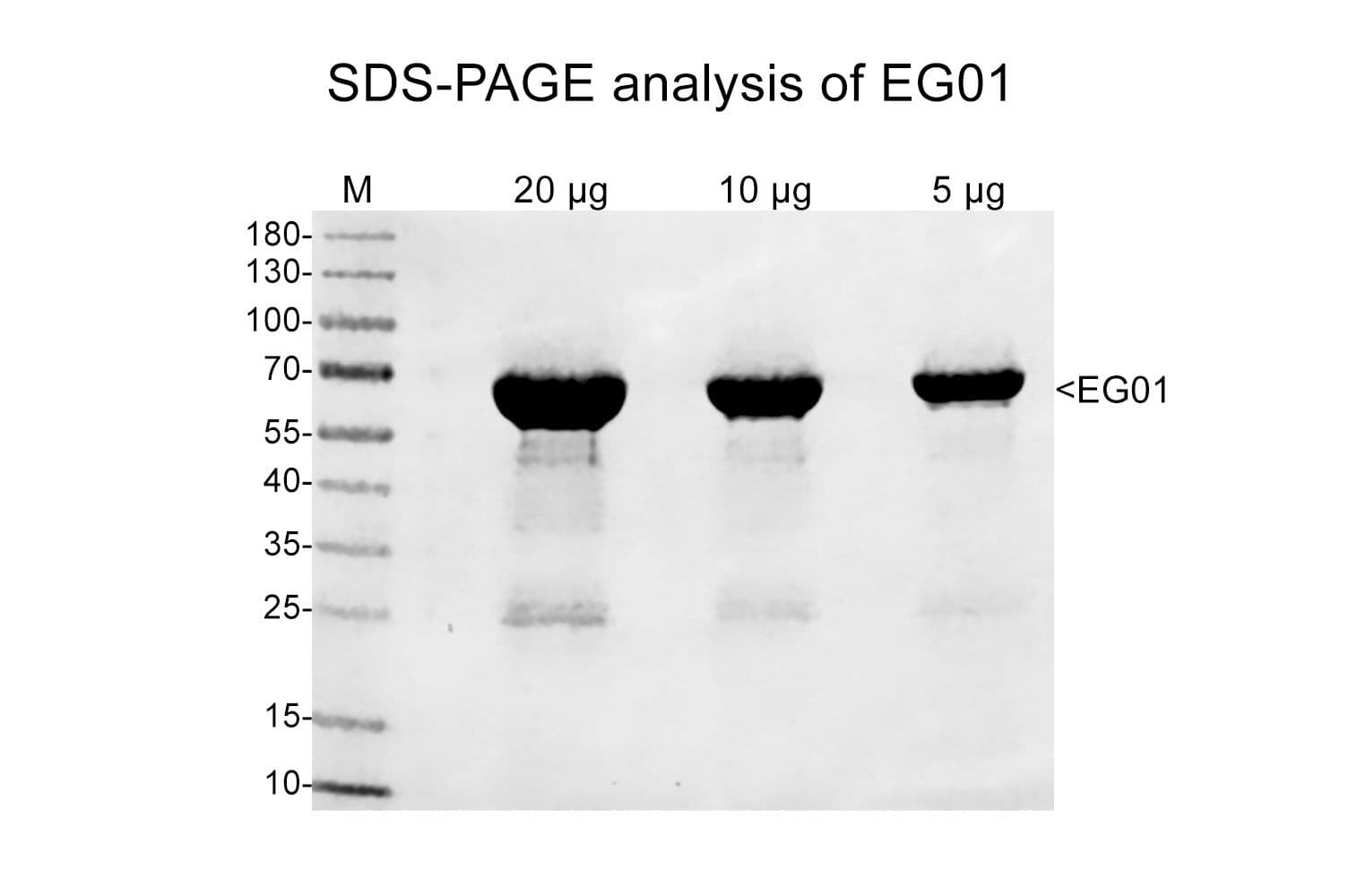
The wild-type human motor domain of KIF11 is produced in a bacterial expression system.
- Protein tag: N-terminal GST-tag
- Molecular weight: 74 kDa
- Motor amino acids: 13-437
- Uniprot ID: P52732
- Formulation: supplied as lyophilized powder
- Composition: when reconstituted 80 mM PIPES pH 6.8, 100 mM NaCl, 2 mM MgCl2, 5% (w/v) sucrose, and 1% (w/v) dextran