September 2019 Newsletter: Membrane Tension and Actin Cytoskeleton Interact to Regulate Cell Motility
Plasma membrane tension regulates many cellular functions, including exocytosis, endocytosis, division, and motility1-6, all of which require a re-shaping of cellular morphology, which in turn relies upon an interplay between plasma membrane deformation and remodeling of the actin cytoskeleton1,3,5-9. This newsletter discusses coordinated interactions between the actin cytoskeleton and membrane tension during motility-associated morphogenesis.
Directed cell motility requires dynamic re-organization of the actin cytoskeleton at the front and rear of the cell (leading edge and trailing edge, respectively) with actin-based lamellipodial protrusions extending and pulling the cell forward in parallel with retraction at the trailing edge via changes in adhesion sites and RhoA-mediated actomyosin contractility8,10,11. As actin-based protrusions push the leading edge of motile cells forward and there is contraction at the trailing edge, the plasma membrane is deformed, changing membrane tension. Directed motility relies upon a balance between actin cytoskeleton remodeling and changes in membrane tension due to deformation8 (Fig. 1).
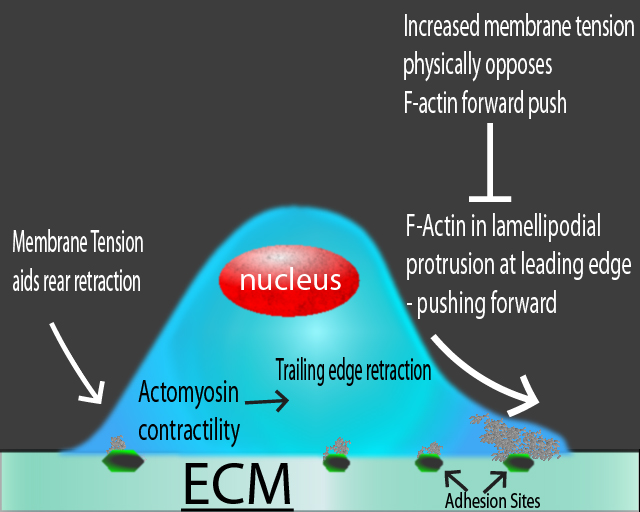
Figure 1. Schematic diagram of different forces in a migrating cell. Actin-based protrusions (i.e., lamellipodia) at the leading edge push the cell forward, while membrane tension physically opposes this motility. At the trailing edge, membrane tension aids in actomyosin-mediated contraction.
One model of how the dynamic re-organization of both membrane tension and actin cytoskeleton interact to enable cell morphogenesis and motility emerged from early 2D cell motility experiments with either lamellipodial fragments from fish keratocytes or living, intact keratocytes8,12. The model hypothesizes that the polymerizing actin network within the leading edge of a motile cell pushes on the cell membrane from within, generating membrane tension that physically opposes the actin-based protrusions, rapidly equilibrates, and exerts globally constant force, per unit filament length, on the polymerizing actin network12 (Fig. 1). At the center of the leading edge, high actin filament density results in low membrane tension per filament, allowing actin filaments to polymerize rapidly and drive protrusion of the leading edge. As the filament density decreases towards the cell’s sides, resistance per filament increases until the membrane tension-induced load stalls actin polymerization, thereby establishing the front corners of the cell12. Similarly, an earlier study in fibroblasts found that membrane tension and lamellipodial protrusion numbers are inversely related1. Concomitantly, the trailing edge of the migrating cells has a disassembling actin network whose depolymerization is hastened by increased membrane tension, resulting in trailing edge retraction12-14 (Fig. 1). Live cell imaging studies of motile keratocytes reported that both in-plane tension (tension in the membrane itself) and membrane-cytoskeletal attachment (adhesions and actomyosin-mediated contractility) regulate membrane tension in motile cells15. Indeed, total membrane tension is determined by both factors8. Lieber et al.15 also confirmed that as actin polymerization increases at the leading edge, the stronger forward propulsion of filaments evokes higher membrane tension and vice versa. At the trailing edge, membrane tension is inversely correlated with myosin contraction levels and directly correlates with adhesion strength15.
Recent studies in fibroblasts and fibroblast-like cells further clarify the interplay between the actin cytoskeleton and membrane tension. A high resolution microscopy study16 in fibroblasts reveals that at the earliest stage of lamellipodial protrusion when the membrane reservoir is sufficient, the length of the protrusion is significant and membrane tension is low. The lamellipodia push outward on the membrane, membrane tension is increased (in-plane tension), and the extending protrusions smooth out any folds and curves in the plasma membrane. As the membrane reservoir is exhausted, tension rapidly increases and concomitantly the actin cytoskeleton is reorganized, resulting in decreased protrusion length16. In neutrophils, increased protrusions at the leading edge of a migrating cell coincide with increased membrane tension and decreased protrusion numbers at cellular locations other than the leading edge7. Another fibroblast study17 reports cyclic oscillations in membrane tension inversely related to lamellipodial protrusion morphology. When tension increases, the protrusions decrease in size (reduced width) and shape (bend upwards) and as tension dissipates, protrusions return to their normal size, shape, and growth activity17. The actin-associated protein, FBP17 (formin-binding protein 17), is a membrane-bending protein and activator of WASP/N-WASP-dependent actin nucleation at the leading edge of migrating COS-1 (fibroblast-like) cells. Membrane tension inhibits FBP17 function by triggering its membrane detachment, providing a signaling pathway by which tension inhibits actin polymerization18,19, suggesting the existence of a negative feedback loop where increased membrane tension eventually inhibits further protrusion development by inactivating pro-actin assembly molecules8,18,19. Other actin binding and capping proteins also regulate the dynamic re-modeling of the actin network that supports leading edge protrusions20. The phospholipase D2 and mammalian target of rapamycin complex 2 mechanosensory signaling cascade in neutrophils also provides a means by which tension inhibits actin nucleation21.
Summary
The functional relevance of membrane tension extends beyond the biophysical realm as membrane tension (and other membrane attributes such as order and potential) impact nearly every vital biological process. Measuring the tension and dynamic changes in living cells has proven an exceedingly difficult undertaking. Challenges include validating the results obtained with 2D cell culture in 3D cell cultures and dissecting how regulation of membrane tension can vary not only between cell types but potentially between the state of the cell when tension is measured8. An exciting development is the development and commercial availability of fluorescent mechanosensitive probes specifically designed to study changes in membrane tension in real-time in living cells9,22,23. Cytoskeleton offers new fluorescence probes to help biophysicists and cell biologists delve deeper into understanding how membrane tension interacts with the cytoskeleton to regulate basic cellular functions. Additionally, Cytoskeleton offers, Flipper-TR™, a new fluorescence probe to help cell biologists delve deeper into understanding how membrane tension interacts with the cytoskeleton to regulate basic cellular functions (Fig. 2). Additionally, Cytoskeleton offers live cell imaging probes for F-actin, microtubules, and DNA, as well as Biochem Kits for studying G-actin and F-actin levels in cells and interaction in vitro.
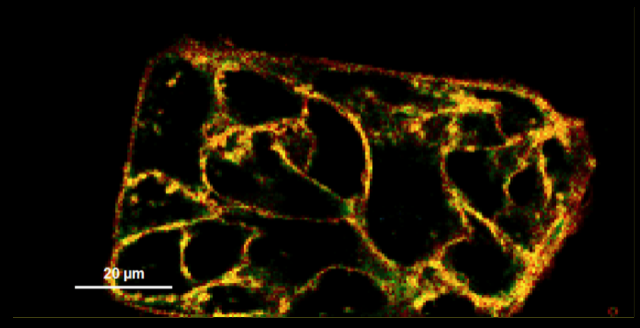
Figure 2: Fluorescent lifetime map of cell membranes stained with FLIPPER-TR membrane tension probe (Cat # CY-SC020). Green represents medium tension, yellow/orange/red is under higher tension, and blue is lower tension. Images courtesy of Colom et al. 2018.
References
- Raucher D. and Sheetz M.P. 2000. Cell spreading and lamellipodial extension rate is regulated by membrane tension. J. Cell Biol. 148, 127-136.
- Keren K. 2011. Cell motility: the integrating role of the plasma membrane. Eur. Biophys. J. 40, 1013-1027.
- Diz-Munoz A. et al. 2013. Use the force: Membrane tension as an organizer of cell shape and motility. Trends Cell Biol.23, 47-53.
- Masters T.A. et al. 2013. Plasma membrane tension orchestrates membrane trafficking, cytoskeletal remodeling, and biochemical signaling during phagocytosis. Proc. Natl. Acad. Sci. U.S.A. 110, 11875-11880.
- Clark A.G. et al. 2014. Stresses at the cell surface during animal cell morphogenesis. Curr. Biol. 24, R484-R494.
- Sens P. and Plastino J. 2015. Membrane tension and cytoskeleton organization in cell motility. J. Phys. Condens. Matter.27, 273103.
- Houk A.R. et al. 2012. Membrane tension maintains cell polarity by confining signals to the leading edge during neutrophil migration. Cell. 148, 175-188.
- Pontes B. et al. 2017. Membrane tension: A challenging but universal physical parameter in cell biology. Semin. Cell Dev. Biol.71, 30-41.
- Colom A. et al. 2018. A fluorescent membrane tension probe. Nat. Chem.10, 1118-1125.
- Warner H. et al. 2019. Control of adhesion and protrusion in cell migration by Rho GTPases. Curr. Opin. Cell Biol.56, 64-70.
- Lawson C.D. and Ridley A.J. 2018. Rho GTPase signaling complexes in cell migration and invasion.J. Cell Biol. 217, 447-457.
- Keren K. et al. 2008. Mechanism of shape determination in motile cells. Nature. 453, 475–480.
- Kozlov M.M. and Mogilner A. 2007. Model of polarization and bistability of cell fragments. Biophys. J. 93, 3811–3819.
- Ofer N. et al. 2011. Actin disassembly clock determines shape and speed of lamellipodial fragments. Proc. Natl. Acad. Sci. U.S.A.108, 20394–20399.
- Lieber A.D. et al. 2013. Membrane tension in rapidly moving cells is determined by cytoskeletal forces. Curr. Biol.23, 1409-1417.
- Gauthier N.C. et al. 2011. Temporary increase in plasma membrane tension coordinates the activation of exocytosis and contraction during cell spreading. Proc. Natl. Acad. Sci. U.S.A. 108, 14467-14472.
- Pontes B. et al. 2017. Membrane tension controls adhesion positioning at the leading edge of cells. J. Cell Biol. 216, 2959-2977.
- Zegers M.M. and Friedl P. 2015. membrane tension into cytoskeletal action by FBP17. Dev. Cell. 33, 628-630.
- Tsujita K. et al. 2015. Feedback regulation between plasma membrane tension and membrane-bending proteins organizes cell polarity during leading edge formation. Nat. Cell Biol.17, 749-758.
- Simon C. et al. 2018. Interplay between membrane tension and the actin cytoskeleton determines shape changes. Phys. Biol.15, 065004.
- Diz-Munoz A. et al. 2016. Membrane tension acts through PLD2 and mTORC2 to limit actin network assembly during neutrophil migration. PLoS Biol. 14, e1002474.
- Molin M.D. et al. 2015. Fluorescent flippers for mechanosensitive membrane probes. J. Am. Chem. Soc. 137, 568-571.
- Goujon A. et al. 2019. Mechanosensitive fluorescent probes to image membrane tension in mitochondria, endoplasmic reticulum, and lysosomes.J. Am. Chem. Soc. 141, 3380-3384.
Related Products
Live Cell Imaging Products
SiR-Actin Kit (Cat. # CY-SC001)
SiR-Tubulin Kit (Cat. # CY-SC002)
Cytoskeleton Kit (Includes SiR-Actin, SiR-Tubulin, and Verapamil) (Cat. # CY-SC006)
SiR-Lysosome Kit (Cat. # CY-SC012)
SiR700-Actin Kit (Cat. # CY-SC013)
SiR700-Tubulin Kit (Cat. # CY-SC014)
SiR700-DNA Kit (Cat. # CY-SC015)
SiR700-Lysosome Kit (Cat. # CY-SC016)
Flipper-TR Kit for fluorescence cell membrane microscopy (Cat # CY-SC020)
BioChem Kits
Actin Binding Protein Spin-Down Assay Biochem Kit: rabbit skeletal muscle actin (Cat # BK001)
Actin Polymerization Biochem Kit (fluorescence format): rabbit skeletal muscle actin (Cat # BK003)
G-Actin/F-actin In Vivo Assay Biochem Kit (Cat # BK037)
G-LISA Kits
RhoA G-LISA Activation Assay (Luminescence format) (Cat. # BK121)
RhoA G-LISA Activation Assay Kit (Colorimetric format) (Cat. # BK124)
RhoA G-LISA Activation Assay Kit (Colorimetric format) (Cat. # BK124-S)
Rac1,2,3 G-LISA Activation Assay (Colorimetric format) (Cat. # BK125)
Rac1 G-LISA Activation Assay (Luminescence format) (Cat. # BK126)
Cdc42 G-LISA Activation Assay (Colorimetric format) (Cat. # BK127)
Cdc42 G-LISA Activation Assay (Colorimetric format) (Cat. # BK127-S)
Rac1 G-LISA Activation Assay Kit (Colorimetric Based) (Cat. # BK128)
Rac1 G-LISA Activation Assay Kit (Colorimetric Based) (Cat. # BK128-S)
RalA G-LISA Activation Assay Kit (Colorimetric Based) (Cat. # BK129)
Ras G-LISA Activation Assay Kit (Colorimetric Based) (Cat . # BK131)
Arf1 G-LISA Activation Assay Kit (Colorimetric Based) (Cat. # BK132)
Arf6 G-LISA Activation Assay Kit (Colorimetric Based) (Cat. # BK133)
RhoA / Rac1 / Cdc42 G-LISA Activation Assay Bundle 3 Kits (Cat. # BK135)
Total RhoA ELISA (Cat. # BK150)
Acti-Stain Phalloidins
Acti-stain 488 Phalloidin (Cat. # PHDG1)
Acti-stain 555 Phalloidin (Cat. # PHDH1)
Acti-stain 670 Phalloidin (Cat. # PHDN1)
Rhodamine Phalloidin (Cat. # PHDR1)
Pull-Down Activation
Ras Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK008)
Ras Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK008-S)
RhoA / Rac1 / Cdc42 Activation Assay Combo Biochem Kit (bead pull-down format) (Cat. # BK030)
Arf1 Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK032-S)
Arf6 Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK033-S)
Cdc42 Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK034)
Cdc42 Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK034-S)
Rac1 Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK035)
Rac1 Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK035-S)
RhoA Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK036)
RhoA Pull-down Activation Assay Biochem Kit (bead pull-down format) (Cat. # BK036-S)
RalA Activation Assay Biochem Kit (bead pull-down format) (Cat . # BK040)
GGA3-PBD Beads (Binds active Arf1 and Arf6 proteins) (Cat. # GGA07)
PAK-PBD beads (binds active Rac/Cdc42 proteins) (Cat. # PAK02)
Rhotekin-RBD beads (binds active Rho proteins) (Cat. # RT02)

